Lithium in Periodic Table
Lithium is a group 1 chemical element or alkali metal in the periodic table that has the symbol Li and atomic number 3. It is a soft, silvery white alkali metal that has the lowest density among all metals. A highly reactive lithium metal reacts vigorously with water and should be stored in a vacuum, an inert atmosphere, or an inert liquid. Lithium was discovered in 1817 by Swedish chemist Johan August Arfvedson when analyzing petalite (LiAlSi4O10) ore. The name of the alkali metal lithium was given from the Greek word ‘lithos’, which means stone. Naturally, it is a mixture of two stable isotopes, Li-6 and Li-7.

Consistent with their ns1 outer electron configuration, the alkali metals occupy group 1 in the periodic table. Lithium was the only alkali metal that was discovered from a mineral, while other common alkali metals were discovered from plant-based material.
Lithium is the third element in the periodic table that contains three protons and three electrons. The pure form of this alkali metal is silvery white and soft, that cut butter with a knife. It does not play any biological role in our body, and it is toxic to human beings.
Lithium carbonate (Li2CO3) is the major commercial form of this metal. It is produced mainly from ores or brines by adding sodium carbonate.
Occurrence of Lithium
Free lithium metal does not occur in nature due to its reactivity. Therefore, small amounts of a combined form are found generally in all igneous rocks and in the waters of many mineral springs.
The most important minerals that contain an extractable amount of lithium are spodumene LiAl(SiO3)2, petalite LiAlSi4O10, lepidolite LiF, LiOH, Al2(SiO3)3, and amblygonite (Li, Na)AlPO4(F, OH).
Until the 1990s, the market of lithium metal was dominated by America, and it was produced from its mineral deposits. However, when we turn on the 21st century, most of the metal is obtained from non-U.S. sources like Australia, Chile, and Portugal. Bolivia contains half the world’s lithium deposits, but it is not a major producer of the metal.
Large deposits of spodumene occur in the USA, Canada, Brazil, Argentina, the Soviet Union, Spain, and Congo. Most of Li metal is currently obtained from brines in Chile. Brines generally yield lithium carbonate when treated with sodium carbonate.
It forms independent minerals because Li+ is too small to replace the more abundant Na+ and K+ in their minerals. It generally occurs in the alumino-silicate minerals along with magnesium. Such minerals generally are separated in the very late stage of crystallization of a magma.
The pure Li metal can be produced by the electrolysis of a molten mixture of lithium chloride (LiCl) and potassium chloride (KCl).
History of Li
The mineral petalite LiAl(Si2O5)2 was discovered in 1800 by the Brazilian statesman José Bonifácio de Andrada e Silva in a mine in Sweden. However, the metal lithium was not known until 1817, before analyzing petalite ore.
The presence of this metal was discovered in 1817 by the Swedish mineralogist Johan August Arfwedson when analyzing petalite ore. He determined that the mineral petalite (LiAISi4O10) contained an unknown element responsible for the crimson color flame.
Arfvedson named the element, but he was unable to purify this alkali metal. Therefore, such an alkali metal was purified or isolated from lithium chloride in 1855 by the German chemists Robert Wilhelm Bunsen and Augustus Matthiessen.
Due to high reactivity, it is not found free in nature. Therefore, it is found naturally as a combined form or compound. Today, a large amount of Li is extracted through the electrolysis of lithium chloride (LiCl).
Extraction of Lithium
Many tonnes of metal are extracted annually throughout the world for different industrial purposes. Besides, several thousand tonnes of lithium salts are produced for various industrial purposes.
The alkali metal lithium is usually extracted and purified from spodumene that contains 1 -3% by following these steps:
- The concentration of Li can increase up to 4 – 6% by oil flotation.
- The α-form of the ore converted to a less dense and more friable β-form by heating to about 1100 °C.
- The β-form can be leached with conc. H2SO4 and water at 250 °C to form soluble Li2SO4. The insoluble silica rendered can be separated by this process.
- Sodium carbonate (Na2CO3) can be added to the solution to form a precipitate of Li2CO3.
- The chloride is formed when we add hydrochloric acid to lithium carbonate. It is used for the production of pure Li metal by electrolysis.
- The electrolysis is generally carried out in a fused mixture of lithium and potassium chlorides. A fused mixture of these two chlorides is used because the melting point of the mixture is lower than that of pure lithium chloride.
- Graphite anode and steel cathode are generally used for the electrolytic production of pure Li. The alkali metal Li is generally liberated at the cathode.
Position of Lithium in Periodic Table
Lithium is placed in group 1 and period 2 of the periodic table. Along a period, it is placed after hydrogen and before sodium metal.
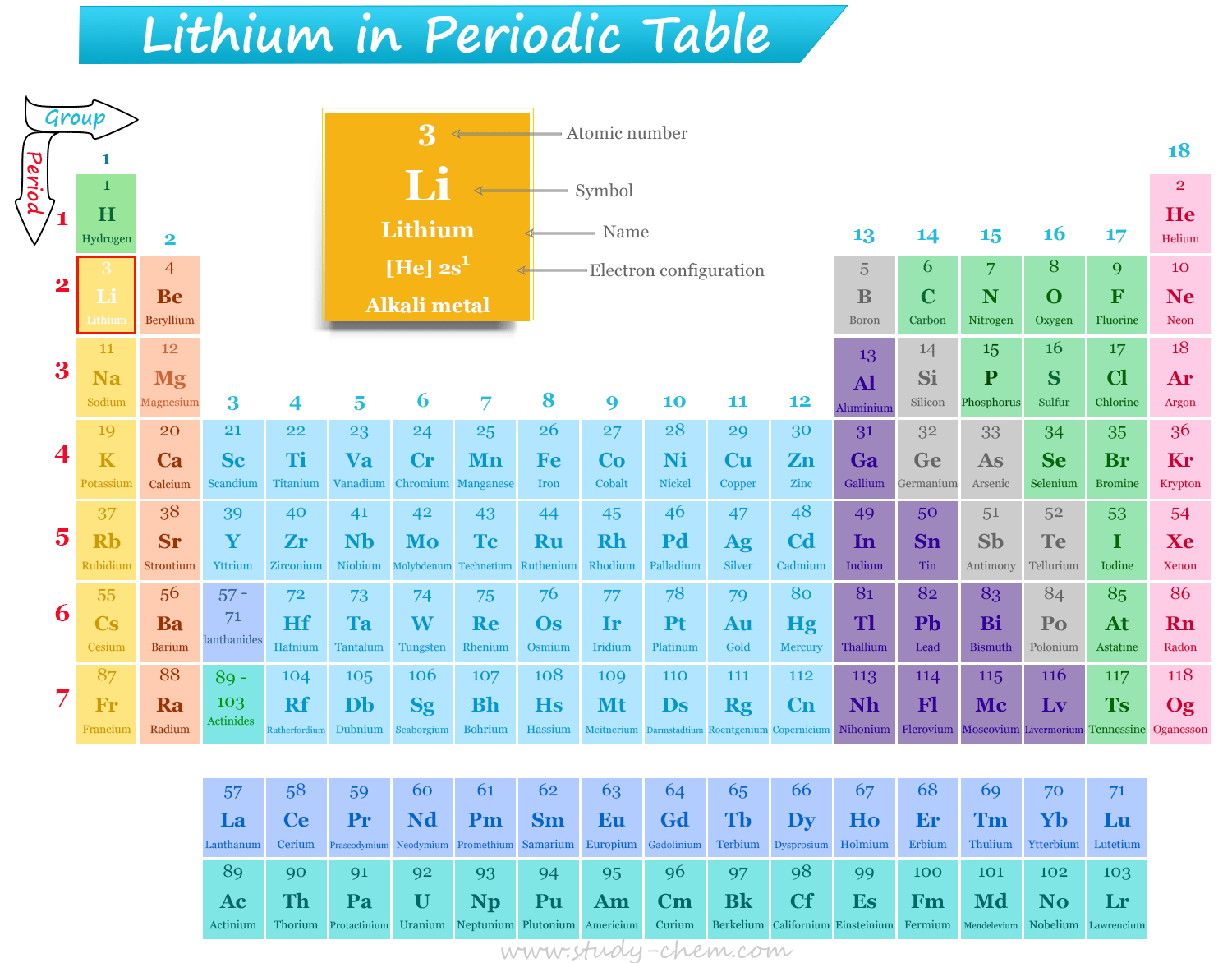
Lithium is one of the alkali metals that is placed in the s-block in the periodic table.
Properties of Lithium
Lithium is the lightest, whitest, and lustrous of the solid chemical elements in the periodic table that forms various industrial alloys and compounds.
| Symbol | Li | ||
| Atomic number | 3 | ||
| Relative atomic mass | 6.94 | ||
| Electron configuration | [He] 2s1 | ||
| Periodic Position | Group 1 and period 2 (s-block) of the periodic table | ||
| Melting point | 180.50°C | ||
| Boiling point | 1342°C | ||
| Density (g cm−3) | 0.534 | ||
| State at 20°C | Solid | ||
| Key isotopes | 7Li | ||
| CAS number | 7439-93-2 | ||
| ChemSpider ID | 2293625 | ||
| Atomic radius, non-bonded (Å) | 1.82 | ||
| Covalent radius (Å) | 1.30 | ||
| Electron affinity (kJ mol−1) | 59.633 | ||
| Electronegativity (Pauling scale) | 0.98 | ||
| Ionisation energies (kJ mol−1) | 1st | 2nd | 3rd |
| 520.222 | 7298.15 | 11815.044 | |
| Common oxidation states | 1 | ||
Chemical Facts
The properties of lithium and its compounds differ significantly from those of the remaining alkali metals due to the small size and very high polarizing power of the Li+ cation. In fact, it resembles magnesium more closely in its behavior due to the diagonal relationship in the periodic table.
The size of Li+ (76 pm) is closer to that of Mg+2 (78 pm) ion. Such data also explains the similar behaviour of Li and Mg.
The major differences of Li from other alkali metals are:
- The alkali metal Li is generally immiscible with molten K, Rb, and Cs and miscible with Na only above 380 °C. However, all the other pairs of alkali metals are freely miscible with each other.
- The solubility of fluoride, hydroxide, phosphate, and carbonates of Li is much less soluble than the corresponding Na and K compounds.
- Li combines directly with nitrogen to form a nitride, while other alkali metals do not form such type of nitride.
- When combined with oxygen, it only forms monoxide (Li2O) while other alkali metals form peroxide and superoxide.
- Lithium sulfate does not form an alum, but sulfates of other alkali metals forms alum.
Chemical Reactivity
Due to the easy excitation of outermost electrons, lithium gives a characteristic flame with crimson colouration. Such a fact has led to the development of analytical methods for the precise estimation of Li by flame photometry and atomic absorption spectroscopy.
The metal Li is highly electropositive, and it has the lowest value of reduction potential among all alkali metals. However, Li has the highest ionization energy among the alkali metals.
The ionization energy is only one of the several factors contributing to the standard electrode potential of an element. Therefore, the electropositive character of lithium metal is explained by its small size and very large hydration energy.
The large difference between the first and second ionization energies of the Li element suggests that the preferred oxidation state of such a metal will be +1.
- Except for nitrogen, the reactivity of the alkali metals increses from lithium to cesium. Therefore, water at 25 °C, it attacks slowly, sodium reacts vigorously, potassium inflames, and rubidium or cesium reacts with explosion. The slow reactivity of Li is due to the kinetic factor.
- The metal Li forms monoxide (Li2O) and a trace amount of peroxide (Li2O2) when burned in air or oxygen.
- The alkali metal Li reacts slowly with nitrogen at room temperature and rapidly at 400 °C to give ruby red crystalline Li3N.
- On heating, Li reacts with carbon to form the dilithium acetylide (Li2C2).
Uses for the Lithium Element
In metallurgy, the active Li metal is used as a scavenger for removing impurities or refining metals such as iron, nickel, copper, and zinc, and their alloys.
Li metal acts as a scavenger and scavenges a large number of nonmetallic elements like oxygen, hydrogen, nitrogen, carbon, sulfur, and the halogens.
- Lithium is the most important component of rechargeable batteries that are commonly used in mobile phones, laptops, digital cameras, and electric vehicles.
- Li metal also plays an important role in making some non-rechargeable batteries for some electronic accessories like heart pacemakers, toys, and clocks.
- Aluminium and magnesium are commonly alloyed with lithium to improve their strength and make them lighter. Such types of alloys are used in armour plating, aircraft making, bicycle frames, and high-speed trains.
- In the glass and ceramic industry, lithium oxide is used for making special glasses and glass ceramics
- Lithium chloride is one of the most important hygroscopic materials that is used in air conditioning systems and industrial drying systems
- Lithium stearate (LiOH + tallow) is used for making high-temperature lubricant greases.
- Lithium carbonate (Li2CO3) is now being used extensively in cells for the extraction of aluminum because it permits larger current flow, reduces fluoride emission, lowers the melting point, and reduces production cost. In the drug industry, it is also used for treating manic depression.
- Lithium hydride (LiH) is a promising storage material that produces hydrogen for use as a fuel. Therefore, it is used to generate hydrogen for military and meteorological requirements.
Frequently Asked Questions
What is lithium?
Lithium is the third element in the periodic table that contains three protons and three electrons. The pure form of the element is so reactive and found naturally as a combined form or compound. Naturally, it is a mixture of two stable isotopes, Li-6 and Li-7.
Who discovered the lithium element?
Lithium was discovered in 1817 by Swedish chemist Johan August Arfvedson when analyzing petalite (LiAlSi4O10) ore. He determined that the mineral petalite (LiAISi4O10) contained an unknown element responsible for the crimson color flame. Arfvedson named the element, but he was unable to purify this alkali metal.
What is lithium used for?
Lithium is used mostly for making rechargeable batteries that are commonly used in mobile phones, laptops, digital cameras, and electric vehicles.
It is also a good alloying metal and forms high-strength, low-density aluminium and magnesium alloys for armour plating, aircraft construction, bicycle frames, and high-speed trains. Many lithium compounds, such as lithium stearate, carbonate, and hydroxide, are used for making greases, the extraction of aluminium, and the extraction of carbon dioxide in space capsules and submarines.
Is Li metal?
Yes, lithium (Li) is an alkali metal that is placed in group 1 and period 2 of the periodic table.
Can you make tritium from lithium?
Yes, lithium may be used to prepare tritium through low-energy nuclear fission.
3Li6 + 0n1 → 1H3 + 2He4 + 4.8 MeV
Tritium is a promising fuel for thermonuclear power generation by fusion.
1H3 + 1H2 → 2He4 + 0n1 + 17.6 MeV
Where is lithium on the periodic table?
Lithium is the third element that is placed in group 1 and period 2 of the periodic table, along with the alkali metals. It is a s-block element that is placed after hydrogen and before sodium.
When was lithium discovered?
The mineral petalite LiAl(Si2O5)2 was discovered in 1800 by the Brazilian statesman José Bonifácio de Andrada e Silva in a mine in Sweden. However, the metal lithium was not known until 1817, before analyzing petalite ore.
The presence of this metal was discovered in 1817 by the Swedish mineralogist Johan August Arfwedson when analyzing petalite ore. However, such an alkali metal was isolated in 1855 by the German chemists Robert Wilhelm Bunsen and Augustus Matthiessen.
What comes after lithium in the periodic table?
Boron comes just after lithium in the periodic table. However, along group1, it is placed after hydrogen and before sodium.
Why is lithium’s symbol Li?
The symbol of the alkali metal lithium Li is derived from the first two letters of its name, which is derived from the Greek word ‘lithos’, which means stone.
What is the main use of lithium carbonate?
A small daily dose of lithium carbonate is used mainly for treating, controlling, preventing, or reducing manic depressive psychoses.
Lithium carbonate (Li2CO3) is now being used extensively in cells for the extraction of aluminum because it permits larger current flow, reduces fluoride emission, lowers the melting point, and reduces production cost. It is also used as a flux in melting porcelain and enamel and in the production of special toughened glass.