Oxidation Number of Periodic Table Elements
Oxidation number of periodic table elements in a compound is the formal charge (positive or negative) which would be assigned to the elements if all the bonds in the compound were ionic bonds. The oxidation numbers are arbitrary because all bonds in the compound are treated as ionic bonds during their calculation. Such arbitrary numbers help in balancing redox reactions, in calculating electron transfer, and in understanding redox chemistry. The concept of electronegativity is utilized in adding a formal charge to an atom. Therefore, the less electronegative partners of a binary compound is arbitrarily assigned a positive oxidation number and the more electronegative one a negative oxidation state.
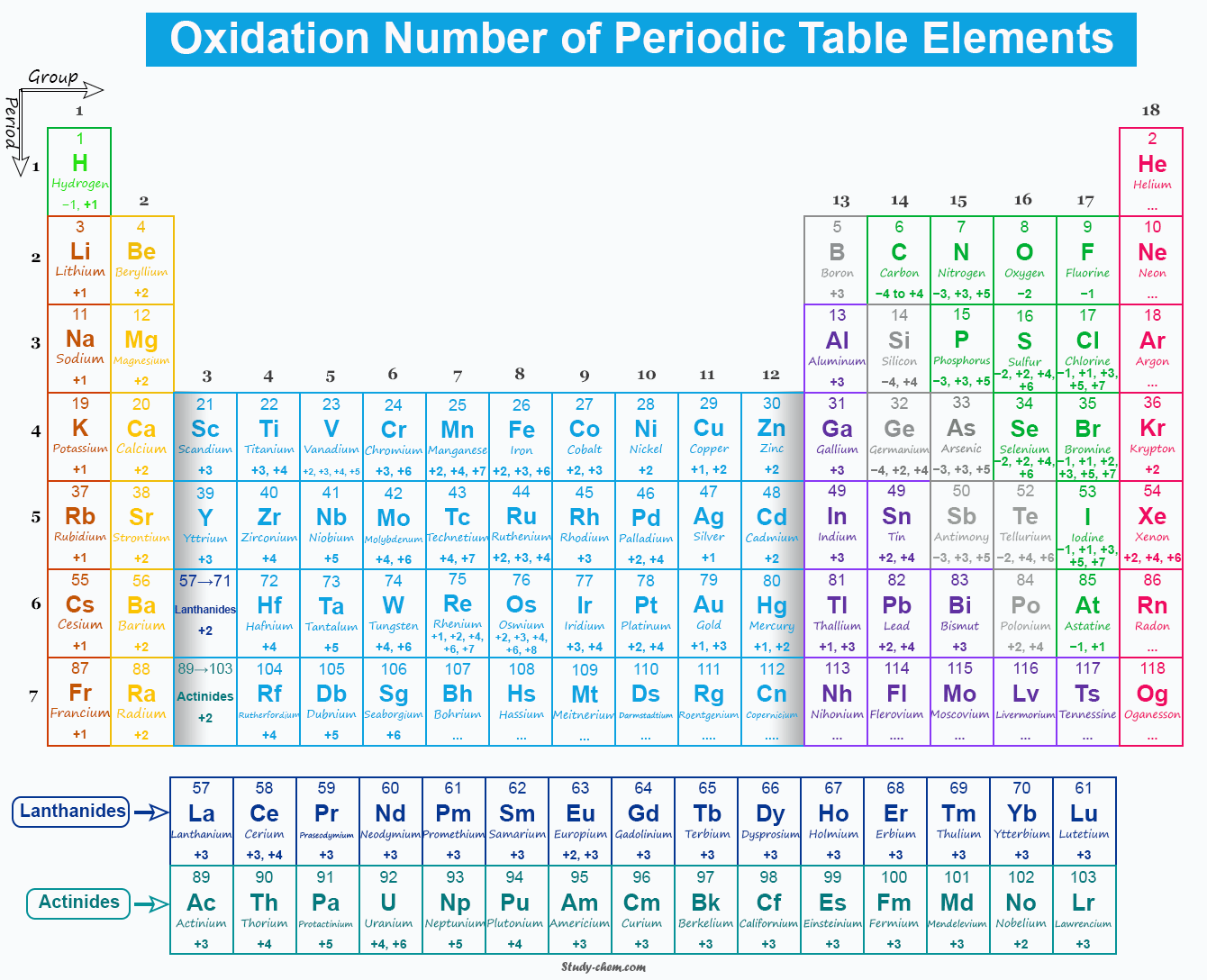
Fluorine is the most electronegative chemical element in the periodic table. Therefore, it always shows a negative oxidation number. However, alkali and alkaline earth metals are the most electropositive elements in the periodic table. Therefore, they always show positive oxidation numbers.
Since all the compounds of alkali and alkaline earth metals have consistently unipositive and bipositive ions. Therefore, they assigned oxidation numbers +1 and +2 respectively. Similarly, halogens and chalcogens generally show -1 and -2 oxidation numbers.
Rules for Calculation of Oxidation Numbers
The following general rules are used for the calculation of oxidation numbers. These rules are:
- The algebraic sum of the oxidation numbers of all the atoms in a compound must be zero.
- The oxidation number of monoatomic ions is equal to their charge.
- The algebraic sum of the oxidation numbers of all the atoms in a polyatomic ions equal to its charge.
- Atoms of diatomic molecules or metallic elements are assigned zero oxidation numbers since the same elements of similar electronegativity are involved in the chemical bonding.
- Oxygen normally has an oxidation number of −2. However, in peroxide and superoxide, the oxidation numbers of oxygen is −1 and −½ respectively.
- Fluorine is the most electronegative element in the periodic table, and it always shows an oxidation number of −1. However, in F2O, the oxidation number of oxygen is +2 because fluorine is more electronegative than oxygen.
- Hydrogen normally has an oxidation number of +1. However, in alkali metal hydrides, the oxidation number of hydrogen is −1.
Examples of Oxidation Number
When the above rules are applied to the calculation of oxidation numbers of carbon in the compounds CH4, CH3Cl, CH2Cl2, CHCl3, and CCl4.
Let the oxidation number of carbon in CH4 be x.
∴ x + 4×(+1) = 0
Or, x = −4
Let the oxidation number of carbon in CH3Cl be x.
∴ x + 3×(+1) + (−1) = 0
Or, x = −2
Let the oxidation state of carbon in CH2Cl2 be x.
∴ x + 2×(+1) + 2×(−1) = 0
Or, x = 0
Let the oxidation number of carbon in CHCl3 be x.
∴ x + (+1) + 3×(−1) = 0
Or, x = +2
Let the oxidation number of carbon in CCl4 be x.
∴ x + 4×(−1) = 0
Or, x = +4
Therefore, the oxidation numbers of carbon in the compounds CH4, CH3Cl, CH2Cl2, CHCl3, and CCl4 are −4, −2, 0, +2, and +4, respectively.
Water (H2O)
H: +1
O: −2
Sum of oxidation number = 2×(+1) + (−2) = 0
Sulfuric Acid (H2SO4)
H: +1
S: +6
O: −2
Sum of oxidation number = 2×(+1) + (+6) + 4×(−2) = 0
Potassium Dichromate (K2Cr2O7)
K: +1
Cr: +7
O: −2
Sum of oxidation number = 2×(+1) + 2(+6) + 7×(−2) = 0
Ferrosoferric oxide (Fe3O4)
Ferrosoferric oxide is a mixed oxide of ferric oxide (Fe2O3) and ferrous oxide (FeO). Therefore, the oxidation state of iron in ferric oxide = +3, and in ferrous oxide = +2.
Ammonium Nitrate (NH4NO3)
Ammonium nitrate (NH4NO3) is formed by ammonium ion (NH4+) and nitrate ion (NO3−). Therefore, the oxidation state of the two nitrogen atoms in NH4NO3 is different.
Let the oxidation state of ammonium nitrogen = x and nitrate nitrogen = y.
Therefore, for NH4+,
x + 4(+1) = 1
Or, x = −3
For NO3−
y + 3(−2) = −1
Or, y = +5
Therefore, the oxidation numbers of the two nitrogen atoms in NH4NO3 are −3 and +5.
Calculation of Oxidation Number
We use the following steps for calculating the oxidation number of an atom in a compound.
- Step 1: Identify the oxidation number of known atoms in a compound. For example, the general oxidation number of oxygen = −2, hydrogen = +1, halogens = −1, alkali metals = +1, and alkaline earth metals = +2.
- Step 2: Let the oxidation number of the unknown atom be x.
- Step 3: Write the algebraic sum of the oxidation numbers of all the atoms in a compound or ion. For a compound, it is equal to zero, but for an ion, it is equal to its charge.
- Step 4: Solve the algebraic equation and find x.
Oxidation number of Mn in KMnO4
The oxidation numbers of K and O in KMnO4 are +1 and −2.
Let the oxidation number of Mn in KMnO4 = x.
∴ +1 + x + 4(−2) = 0
Or, x − 7 = 0
Or, x = +7
Oxidation number of sulfur in SO4−2
The oxidation number of O = −2.
Let the oxidation number of S in SO4−2 = x.
∴ x + 4(−2) = −2
Or, x −8 = −2
Or, x = +6
Chromium in K2CrO4
Oxidation of potassium = +1 and oxygen = −2.
Let the oxidation number of chromium in K2CrO4 = x.
∴ 2(+1) + x + 4(−2) = 0
Or, x −6 = 0
Or, x = +6
Barium in barium peroxide (BaO2)
The oxidation number of peroxo oxygen = −1.
Let the oxidation number of barium in barium peroxide = x.
∴ x + 2 (−1) = 0
Or, x − 2 = 0
Or, x = +2
Nitrogen in sodium azide (NaN3)
The oxidation number of sodium = +1.
Let the oxidation number of nitrogen = x.
∴ +1 + 3x = 0
Or, 3x = −1
Or, x = −1/3
Oxidation number of P in H4P2O7
Let the oxidation number of P in H4P2O7 = x
∴ 4(+1) + 2x + 7(−2) = 0
Or, 2x = +10
Or, x = +5
Oxidation number of both chlorine in calcium oxychloride, Ca(OCl)Cl
Ca(OCl)Cl molecule forms by one Cl− ion and one OCl− ion. Therefore, for Cl− ion, oxidation number = −1.
Let the oxidation number of the other chlorine atom in OCl− ion = x.
∴ −2 + x = −1
Or, x = +1
Therefore, the oxidation numbers of one chlorine atom in Ca(OCl)Cl is −1, and the other chlorine atom is +1.
Exceptional Oxidation State
Oxygen generally shows a −2 oxidation state, but in many compounds it shows an exceptional oxidation state. For example, in peroxide and superoxide, the oxidation number of oxygen is −1 and −½ respectively. Similarly, in F2O, the oxidation state of oxygen is +2 because fluorine is more electronegative than oxygen.
Similarly, hydrogen generally shows a +1 oxidation state, but in many compounds it shows an exceptional oxidation state. For example, in metal hydrides such as NaH, CaH2, LiAlH4, the oxidation state of hydrogen = −1.
Chromium in CrO5
The structure of the CrO5 molecule shows that it contains two peroxo bonds. Therefore, in CrO5, the oxidation state of four oxygen atoms = −1 and one oxygen atom = −2.
Let the oxidation state of Cr in CrO5 = x.
∴ x + 4(−1) + 1(−2) = 0
Or, x = +6
Sulfur in Na2S2O3
The structure of Na2S2O3 shows that one sulfur is bonded to another sulfur atom by a coordinate covalent bond or dative bond. Therefore, the oxidation state of the two sulfur atoms in Na2S2O3 is different. The oxidation state of the sulfur atom, which accepts two electrons = −2.
Let the oxidation state of the other sulfur atom = x.
∴ 2(+1) + x + (−2) + 3(−2) = 0
Or, x = +6
Oxidation and Reduction Reactions
The definition of oxidation and reduction based on loss or gain of electrons is limited because such a definition holds good only for ionic compounds. For example, the formation of water from H2 and O2 can not be covered by the electronic concept because water is not an ionic compound. Classically, we can say that hydrogen is oxidized to form water molecules.
In order to cover all oxidation and reduction reactions, or redox reactions, the oxidation number concept has been developed. It can easily identify oxidation or reduction reactions by the increase and decrease of oxidation numbers.
Oxidation Reactions
According to the oxidation number concept, oxidation is a process in which an increase in oxidation number occurs. Let us explain it by the formation of water from hydrogen and oxygen, and the formation of magnesium chloride from magnesium and chlorine.
H2 + O2 → H2O
Mg + Cl2 → MgCl2
The oxidation numbers of hydrogen in a molecular state is zero, but in water it is +1. Similarly, magnesium in elementary state has a zero oxidation state, but in MgCl2 it is +2.
During the formation of water from hydrogen and magnesium chloride from magnesium, the oxidation number of hydrogen and magnesium increses. Therefore, oxidation of hydrogen and magnesium occurs during the formation of water and magnesium chloride.
Reduction Reactions
According to the oxidation number concept, reduction is a process in which a decrese in oxidation number occurs. Oxidation and reduction are always found to go hand in hand during a redox reaction. Therefore, when an element or compound is oxidized, another element or compound must be simultaneously reduced.
In the above two reactions, the oxidation numbers of oxygen decrease from 0 to −2, and chlorine decreases from 0 to −1. Therefore, reduction of oxygen and chlorine occurs during the formation of water and magnesium chloride.
An oxidant is reduced, and simultaneously the reductant is oxidized. Therefore, in the above two reactions, the oxidants oxygen and chlorine can be reduced, and the reductants hydrogen and magnesium can be oxidized.