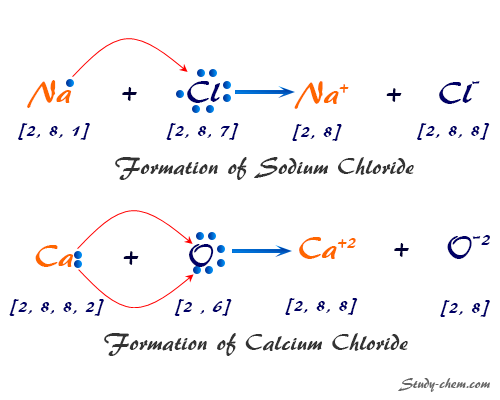
What is Electronic Configuration?
Electronic configuration or electronic structure or electron configuration of periodic table elements is the arrangement of electrons in orbitals around an atomic nucleus. It is a symbolic notation by which the electrons of its atoms are distributed over different atomic orbitals. Electronic configuration is very useful for predicting the valency of an element and the general chemical properties of the periodic table elements. The distribution of electrons in atomic orbitals came into practice shortly after the Bohr model of the atom was presented by Niels Bohr in the year 1913.

Electronic configurations of atoms follow a standard notation in which all electron-containing atomic subshells are placed in a sequence. The number of electrons held in each orbital is written in superscript. For example, the electron configuration of magnesium (atomic number 12) is 1s2 2s2 2p6 3s2.
Elements with a relatively large atomic number have lengthy electron configurations. In such cases, the electronic configuration of completely filled subshells of a noble gas is replaced with the symbol of that noble gas in square brackets. Therefore, the abbreviated electron configuration of magnesium is [Ne] 3s2 (the electron configuration of neon is 1s2 2s2 2p6).
Rules for Writing Electron Configuration
Writing the electronic configuration or electron configuration of atoms of periodic table elements in their respective orbitals can follow some general rules. Such rules for writing electronic configuration are illustrated below:
Shells and Subshells
The maximum number of electrons in the main quantum shell = 2n2, where n = principal quantum number. The main quantum shell, values of n, and the total number of electrons are given below in the table:
| The main quantum shell and n value | Total number of electrons |
| K shell, n=1 | 2 × 12 = 2 |
| L shell, n=2 | 2 × 22 = 8 |
| M shell, n=3 | 2 × 32 = 18 |
| N shell, n=4 | 2 × 42 = 32 |
Principal quantum number or main shell is divided into sub-shells or orbitals like s, p, d, f, etc. The maximum number of electrons in a subshell = 2(2l +1), where l = 0, 1, 2, 3 for s, p, d, and f orbitals respectively. Therefore, s, p, d, and f orbitals can have a maximum of 2, 6, 10, and 14 electrons respectively.
| The main quantum shell and n value | Value of l and sub-shell designation (nl) | The maximum number of electrons in the orbital |
| K shell, n=1 | l = 0 (1s) | 2 |
| L shell, n=2 | l = 0 (2s) | 2 |
| l = 1 (2p) | 8 | |
| M shell, n=3 | l = 0 (3s) | 2 |
| l = 1 (3p) | 8 | |
| l = 2 (3d) | 10 | |
| N shell, n=4 | l = 0 (4s) | 2 |
| l = 1 (4p) | 6 | |
| l = 2 (4d) | 10 | |
| l = 3 (4f) | 14 |
Aufbau Principle
Aufbau building up principle provides the electron filling up process. Hence, according to this principle, the orbitals are filled up in the order of increasing energy. Therefore, orbitals with the lowest energy are filled up first, while the highest energy orbitals are filled up in the end.
Energy Level Diagram
The energy of various subshells or orbitals increses in the order given below:
1s < 2s < 2p < 3s < 3p < 4s < 3d < 4p < 5s < 4d < 5p < 6s < 4f < 5d < 6p < 7s < 5f…
Such an order can also be shown in a diagram. It is called an orbital energy level diagram. However, it is difficult for the students to remember the orbital energy level diagram according to the increasing energy level.
Therefore, a trivial and more convenient diagram was replaced by it for easy remembering. This diagram is generally called an electron configuration diagram.
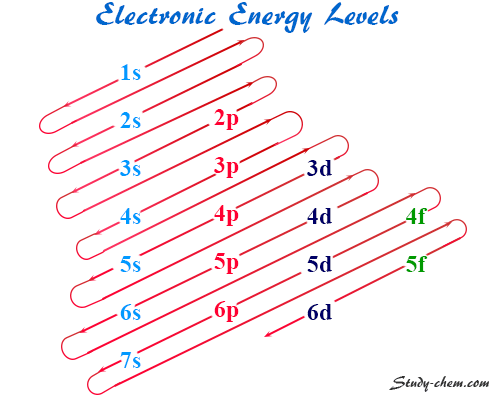
A working rule also proposed how a new electron enters the orbitals. This rule states that a new electron enters the orbital in which (n+l) is minimum.
Therefore, if we consider 3d and 4s orbitals, the electron will first enter the 4s orbital then the 3d orbital because the value of (n+l) in the 4s orbital (4+0 = 4) is less than that of the 3d orbital (3+2 = 5).
However, when the orbitals in which (n+l) values are the same. In such cases, the new electron enters the orbital in which the principal quantum number (n) is minimum.
For example, if we consider 3d and 4p orbitals, the (n+l) values of both the orbitals are the same.
- For 3d orbital (n+l) = 3+2 = 5
- For 4p orbital (n+l) = 4+1 = 5
In such cases, the electron will prefer to go in the 3d orbital because n is lower for the 3d orbital.
Pauli Exclusion Principle
The distribution of electrons in an orbital is governed by the Pauli exclusion principle. Such a principle states that it is impossible for two electrons in a given atom to have all four quantum numbers identical. Therefore, two electrons in the same orbital should have opposite spins because the spin quantum numbers are not identical.
Each of the sub-orbitals (s, px, py, pz, dxy, dxz, dyz, etc) can hold only two electrons, and these two electrons must have opposite spins.
Hund’s Rule
Hund’s put an empirical rule known after this name as Hund’s rule of maximum multiplicity. This rule states that electrons enter the sub-orbitals in such a way as to give the maximum number of unpaired electrons.
When several sub-orbitals of equal energy or degenerate orbitals are available, electrons prefer to occupy separate orbitals rather than pair in the same orbital. Therefore, pairing begins with the introduction of the second electron in the s-orbital, fourth in the p-orbital, and sixth in the d-orbital, and eighth in the f-orbital.
Writing Electronic Configuration of Atom
The electronic configuration of an atom is written in terms of nlx notation.
Where n = main energy shell number,
l = sub-shells or orbitals such as s, p, d, f…,
x = total number of electrons present in the subshell
For example, when we consider oxygen, the atomic number of oxygen is 8. Therefore, it holds 2 electrons in the K shell (n = 1) and 6 electrons in the L shell (n = 2).
The K shell has only the 1s orbital to hold the two electrons while the L shell has 2s and 2p orbitals to hold the six electrons. Therefore, the electronic configuration of oxygen according to the above discussion is 1s2 2s2 2p4.
Magnesium Electronic Configuration
Magnesium has an atomic number of 12 and 12 electrons are distributed in the following manner:
- K shell, n=1 (1s): 2 electrons distributes as 1s2
- L shell, n = 2 (2s, 2p): 8 electrons distributed as 2s2 2p6
- M shell, n = 3: Remaining 2 electrons distributed as 3s2
Therefore, the electron configuration of magnesium according to the above discussion is 1s2 2s2 2p6 3s2 or as [Ne] 3s2.
Electronic Configuration of Periodic Table Elements
The electronic configuration of atoms of the periodic table elements in the ground state are determind with the help of the Aufbau Principle, Hund’s Rule, and Pauli’s Exclusion Principle. According to the electronic configuration, the periodic table elements are classified into four blocks, such as s, p, d, and f-block elements.
Electronic Configuration of s-block Elements
The s-block elements consist of one electron or two electrons in the outer ns-orbital. Therefore, the elements of group-1 and group-2 belong to this block. The general valence shell electronic configuration of s-block elements is ns1→2.
Group 1 Elements
The elements, hydrogen, lithium, sodium, potassium, rubidium, and francium belong to group 1 in the periodic table. The general valence shell electronic configuration of such periodic table elements is ns1.
| Atomic Number | Chemical Elements | Electron Configuration |
| 1 | Hydrogen (H) | 1s1 |
| 3 | Lithium (Li) | 1s2 2s1 |
| 11 | Sodium (Na) | [Ne] 3s1 |
| 19 | Potassium (K) | [Ar] 4s1 |
| 37 | Rubidium (Rb) | [Kr] 5s1 |
| 55 | Cesium (Cs) | [Xe] 6s1 |
| 87 | Francium (Fr) | [Rn] 7s1 |
Group 2 Elements
The elements beryllium, magnesium, calcium, strontium, barium, and radium belong to group 2 in the periodic table. The general valence shell electronic configuration of such group 2 elements = ns2.
| Atomic Number | Chemical Elements | Electronic Configuration |
| 4 | Beryllium (Be) | 1s2 2s1 |
| 12 | Magnesium (Mg) | 1s2 2s2 |
| 20 | Calcium (Ca) | [Ne] 3s1 |
| 38 | Strontium (Sr) | [Ar] 4s1 |
| 56 | Barium (Br) | [Kr] 5s1 |
| 88 | Radium (Ra) | [Xe] 6s1 |
Frequently Asked Questions
What is the electronic configuration of copper?
The atomic number of copper is 29, and the electronic configuration of the copper atom is 1s2 2s2 2p6 3s2 3p6 4s1 3d10 or [Ar] 4s1 3d10.
Such an electronic configuration does not obey aufbau principle because the energy gap between the 3d and the 4s orbital is very small, and a completely filled d-orbital has greater stability than the partially filled d orbital.
Why is the electronic configuration important?
Electronic configuration is very useful and important for predicting the valency of an element and the general chemical properties of the periodic table elements. It also helps to classify periodic table elements into four blocks, such as s, p, d, and f-block elements. Therefore, it is very useful for the collective study of the periodic table elements.
How to write the electronic configuration of noble gases?
The electronic configuration of noble gases is written by following way:
| Name and Symbol | Atomic number | Electronic configuration |
| Helium (He) | 2 | 1s2 |
| Neon (Ne) | 10 | [He] 2s2 2p6 |
| Argon (Ar) | 18 | [Ne] 3s2 3p6 |
| Krypton (Kr) | 36 | [Ar] 3d10 4s2 4p6 |
| Xenon (Xe) | 54 | [Kr] 4d10 5s2 5p6 |
| Radon (Rn) | 86 | [Xe] 4f14 5d10 6s2 6p6 |
| Oganesson (Og) | 118 | [Rn] 5f14 6d10 7s2 7p6 |