Boron in Periodic Table
Boron is a group 13 and period 2 periodic table element that has the chemical symbol B and atomic number 5. It is a high melting solid which found in nature in two forms, crystalline (black) or amorphous (brown). The pure crystalline form of boron is hard and brittle. It is a diamagnetic substance with semiconductor properties. Such a metalloid is naturally present in plants and is essential for the plant’s cell walls. The metalloid, boron, and its compounds are used extensively in the modern age, from making many daily-use items to fertilizers. Boron is the first element of group 13 and the 5th perodic table element that has [He] 2s22p1 electronic configuration.

Boron is an essential nutrient for plants but not poisonous to animals. However, a higher dose can upset the body’s metabolism. It is a trace element in humans but an essential micronutrient of plants. The human body generally intakes 2 milligrams of B in its daily diet. A requisite amount of boron is present in raisins, almonds, prunes, and hazelnuts.
Natural form of this element consist two isotopes, 10B and 11B. Along with these, it has 11 synthetic isotopes with mass numbers ranging from 7 to 17.
Occurrence
The occurrence of boron in our universe is very low. It occurs in our nature as borates in the minerals borax, Na2[B4O5(OH)4]8H2O, and colemanite, Ca2[B3O4(OH)3]22H2O. It also occurs in the form of orthoboric acid in some volcanic spring waters. A small quantity of boron is found in groundwater due to leaching from soils and rocks containing borosilicates and borates.
A huge deposit of borax is found in Turkey, Russia, and Argentina. A precipitate form of borax also occurs in India, Tibet, and Sri Lanka. However, rasorite, Na2[B4O6(OH)2]3H2O is the most important source of boron, which is found in the Mojave Desert in California, USA.
History and Discovery
Borax, Na2B2O5(OH)4 was used as a flux and to prepare glazes and hard glasses in India, Egypt, and other ancient civilizations.
The element itself was isolated in 1808 by Louis-Josef Gay-Lussac and Louis-Jacques Thénard working in Paris, and Sir Humphry Davy in London. They independently extracted it by heating borax with potassium metal.
However, they can not produce the pure form of this element because it is very difficult to purify. In 1892, French chemist Henri Moissan extracted a purer form of this element. However, in 1909, American chemist E. Weintraub successfully produced a pure crystalline form of this element (>99.9%) in the USA by sparking boron chloride vapor and hydrogen.
Extraction Process
A high purity (> 99.9%) of crystalline form is obtained by reduction of volatile boron compounds such as BCl3 or BBr3 with hydrogen on a heated tantalum wire. However, such an extraction process operates only in kilogram scale.
- Below 1000 °C, an amorphous form of this element is obtained.
- α and β-rhombohedral B is obtained between 1000 and 1200 °C.
- However, above 1200 °C, tetragonal crystals of B are formed.
A large quantity of amorphous boron is obtained when reducing the oxide of this metalloid (B2O3) with magnesium (Mg) metal or other electropositive elements at high temperature.
B2O3 + 3Mg → 2B + 3MgO
The unreacted substances and magnesium oxide (MgO) can be removed by washing with dilute hydrochloric acid (HCl) and sodium hydroxide (NaOH) solutions. The boron obtained by this process is usually 95 to 98% pure.
A 95% pure powdered form of boron can be obtained cheaply by electrolytic reduction of KBF4 in molten KCl/KF at 800 °C.
Properties of Boron
Boron is the first element of group 13 in the periodic table that has a very small size and very high electronegativity. Such facts control the properties of boron to a great extent. Therefore, the chemistry of B is slightly different from that of other elements in this group.
| Symbol | B | ||
| Discovery | Discovered in 1808 by Louis-Josef Gay-Lussac and Louis-Jacques Thénard in Paris, France, and Humphry Davy in London, UK. | ||
| Origin of the name | The name originates from the Arabic ‘buraq’, which was the name for borax. | ||
| Relative atomic mass | 10.81 | ||
| Atomic number | 5 | ||
| Electron configuration | [He] 2s22p1 | ||
| CAS number | 7440-42-8 | ||
| ChemSpider ID | 4575371 | ||
| Periodic Position | Group 13 and period 2 (p-block) periodic table | ||
| Allotropes | α-rhombohedral B, β-rhombohedral B, γ-B, and tetragonal boron. | ||
| Key isotopes | 11B | ||
| Appearance at 20°C | Pure form is a brittle, dark, lustrous solid powder. | ||
| Melting point | 2077°C | ||
| Boiling point | 4000°C | ||
| Density (g cm−3) | 2.34 | ||
| Atomic radius, non-bonded (Å) | 1.92 | ||
| Covalent radius (Å) | 0.84 | ||
| Electron affinity (kJ mol−1) | 26.989 | ||
| Electronegativity | 2.04 (Pauling scale) | ||
| Ionisation energies (kJ mol−1) | 1st | 2nd | 3rd |
| 800.637 | 2427.069 | 3659.751 | |
| Common oxidation states | 3 | ||
| Specific heat capacity (J kg−1 K−1) |
1026 | ||
Chemical Properties
The electronic configuration of boron suggests that it is restricted to the small compact 2p orbital. The next available orbital is the 3s orbital, and it is separated from the 2p level by a considerable energy gap. Therefore, this element can not use the 3s orbital for chemical bonding.
Such a periodic table element generally uses two 2s electrons and one 2p electron for covalent chemical bonding. It is chemically inert to air oxygen at normal temperature. However, at high temperatures, it reacts with oxygen to form its oxide.
Generally, it does not react with water but reacts vigorously with halogens to form boron halides.
Interesting Facts
- Boron is the only metalloid of group 13, consisting of other elements such as aluminum, gallium, indium, and thallium.
- Boron is the only element in group 13 that shows a high melting point. However, other elements in this group are low-melting and soft metals.
- Due to the formation of only three covalent bonds with other periodic table elements, the central B atom is electron-deficient.
- Crystalline pure elemental form this element is chemically very inert, and it is not affected by acids or any other oxidizing agents.
Periodic Table of Elements Boron
The atomic number of boron is 5, and the electronic configuration of the element is [He] 2s22p1. Therefore, a brittle, dark, lustrous metalloid boron is positioned in group 13 and period 2 of the periodic table. The valence shell electronic configuration also suggests that it is a p-block element, which is placed after beryllium and before carbon in the periodic table.
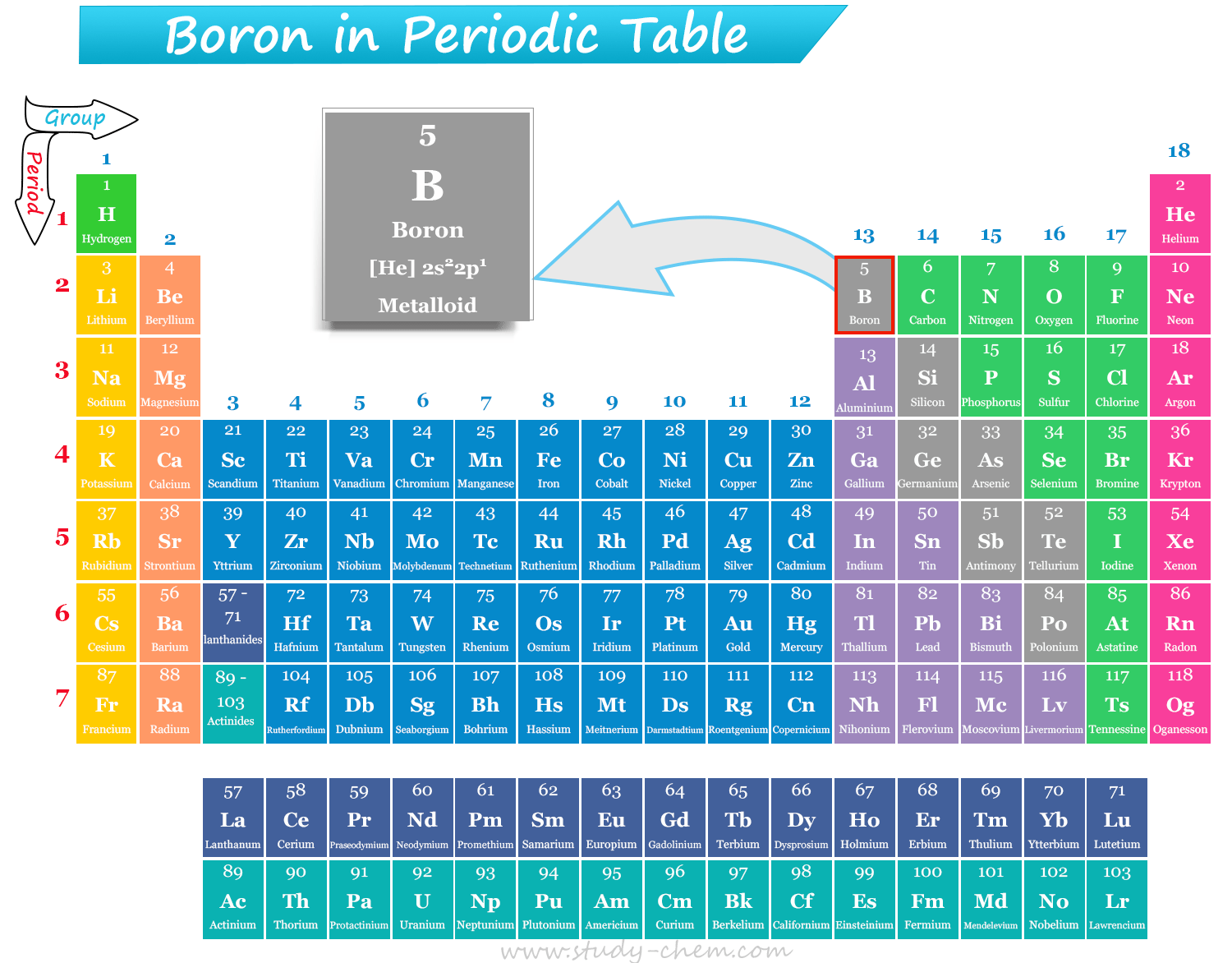
It is the 5th element, or first element of group 13 of the periodic table. Therefore, B is placed along with other group 13 elements: aluminium (Al), gallium (Ga), indium (In), thallium (Tl), and nihonium (Nh).
Uses of Boron
The metalloid, boron, and its compounds are used extensively in the modern age for making many daily-use items to fertilizers. The compounds of boron, such as boric (or boracic) acid, borax (sodium borate), and boric oxide, are extensively used in the modern age. Such compounds are used for making eye drops, mild antiseptics, washing powders, and tile glazes.
- Pure amorphous boron is used as a rocket fuel igniter and pyrotechnic flare with a distinctive green colour.
- Due to the hard and high melting point of metal borides, they are used for coating on turbine blades, rocket nozzles, and high-temperature reaction vessels.
- Borax is an important compound of boron that is used widely for bleaching and preservation of foods.
- Boric acid is an important component of various types of medicines, such as eye drops and mild antiseptics.
- Boric oxide is used for the manufacture of tough and heat-resistant borosilicate glass (Pyrex).
- The compounds, boron carbides and metal borides, are used widely as neutron shields and control rods in nuclear reactors. Boron carbide is also used for polishing and grinding.
- The isotope B-10 is a good absorber of neutrons. Therefore, recently, it has been used in neutron capture therapy of brain tumours.
- The compound, sodium octaborate, is a good flame retardant.
- It is an important component in semiconductor doping, and boron-doped diamond is an excellent material for electrodes.
- B alloyed with steel to form a hard boron steel that prevents wear and tear.
Frequently Asked Questions
What is boron?
Boron is a group 13 and period 2 periodic table element that has the chemical symbol B and atomic number 5. It is a high melting solid that is found in nature in two forms, crystalline (black) or amorphous (brown). The pure crystalline form of B is hard and brittle.
Is boron a metal, nonmetal, or metalloid?
Boron is not a metal or nonmetal. It has properties that are intermediate between metals and nonmetals, and it is called a metalloid.
Who and when was boron discovered?
Such periodic table elements were discovered in 1808 by French chemists, Louis-Joseph Gay-Lussac and Louis-Jacques Thénard in Paris, France, and English chemist Humphry Davy in London, UK. The name of this element is derived from the Arabic word ‘buraq’, a term signifying the element.
What is boron used for?
The metalloid, boron, and its compounds are used extensively in the modern age for making many daily-use items and fertilizers. The compounds of this element, such as boric (or boracic) acid, borax (sodium borate), and boric oxide, are extensively used in the modern age.
Such compounds are used for making eye drops, mild antiseptics, washing powders, and tile glazes.
In everyday life, the compounds, sodium borate or borax, are important ingredients for making eye drops, washing powders (laundry detergents), mild antiseptics, tile glazes, food and wood preservatives, and bleach. In agriculture, boron compounds are used for making various organic fertilizers, insecticides, and pesticides.
What does boron do for the body?
Boron is an essential nutrient for plants but not poisonous to animals. It is a trace element for human beings. However, a higher dose can upset the body’s metabolism.
Various supplements of boron are useful medicines that help to treat painful menstruation and increase sex hormone testosterone levels. Such supplements are also useful for treating osteoarthritis and rheumatoid arthritis, depression, build strong muscles and bones.
Boron is beneficial for post-menopausal women and healthy men because it increases estrogen levels in such women and men.
Such a group 13 element also reduces skin aging, inflammation, brain fog, hair loss (it promotes hair growth), fungal infections, etc. The deficiency of this element may cause osteoporosis and psoriasis.
Is boron dangerous to health?
Boron is a trace element for human beings, and the pure form is not known to be harmful. However, too much intake of boron supliments are hazardous to the body’s metabolism.
A small ammount b is considered safe during pregnancy and breastfeeding. It is always best to consult with your doctor before taking boron supplements.