Carbon Element in Periodic Table
Carbon is a unique, nonmetallic, tetravalent element in Group 14 of the periodic table that has the symbol C and atomic number 6. The atomic form of carbon is very short-lived, but it is stabilised by various allotropic forms. A number of pure forms of carbon are graphite, diamond, fullerenes, and graphene. All these forms of this element have different appearances. For example, diamond is a colourless, transparent, crystalline solid and the hardest material on Earth, while graphite is a soft, black, and shiny material. Carbon is unique among all the periodic table elements because it has the ability to form strongly bonded chains with hydrogen and itself.

Carbon is the second most abundant element in the human body, which is essential for the formation of biomolecules like DNA, proteins, carbohydrates, lipids, etc. Naturally occurring carbon has three isotopes with mass numbers 12, 13, and 14. Among these, C-12 is the most abundant, whereas C-14 is a radioactive isotope of this element.
A number of pure and impure forms of carbon have different appearances and different uses. For example, coal, oil, and natural gas are used as sources of fossil fuels, and petroleum products are used in petrochemical industries for producing polymers, fibres, paints, solvents, plastics, etc. The impure forms, like charcoal and coke, are used widely in metal smelting.
Natural Abundance
Carbon is the 4th most abundant element in the universe by mass after hydrogen, helium, and oxygen. It is generally found in the metal carbonates.
Carbon is present in the Earth’s crust to the extent of about 0.02%. Crude oil and coal are the most common free and combined forms of carbon. The pure allotroic forms include diamond, graphite, graphene, and fullerene.
Flakes of graphite occur in the metamorphosed sedimentary rocks, like quartz. Diamonds occur embedded in other rocks associated with ancient volcanic pipes. Weathering and erosion of such pipes, followed by geological action, also lead to their accommodation in alluvial gravels.
India and Borneo are the oldest diamond-producing countries in the world. Diamond was found in Brazil in 1729, while South africa deposit were discovered only in the late part of the nineteenth century. However, a large deposit was discovered in Siberia in the twentieth century.
History and Discovery
The forms of soot, charcoal, graphite, and diamonds have been known since ancient times. Therefore, the discoverer and discovery date of carbon are unknown. Different allotrope of carbon was discovered in diffent times. However, in ancient times, no one could realize that soot, charcoal, graphite, and diamonds were different forms of carbon.
French scientist Antoine Lavoisier identified that diamond and charcoal were different forms of the same element, and he named it carbon. In 1772, Lavoisier and many other chemists placed a diamond in a closed glass jar and focused the sun’s rays on it with a remarkable giant magnifying glass. They saw that the diamond burned and disappeared.
Lavoisier noted that the overall weight of the jar was unchanged after burning and diamond combined with oxygen to form carbon dioxide. After that, he concluded that diamond and charcoal were different forms of the same element, carbon.
Swedish scientist Carl Scheele in 1779 also carried out the same experiment with graphite. He burned graphite to form carbon dioxide and suggested that it is another allotrope of carbon.
English chemist Smithson Tennant in 1796 established that diamond was pure carbon because it forms only carbon dioxide when burning. He also showed that when equal weights of charcoal and diamonds were burned, they form an equal amount of carbon dioxide.
English chemist Benjamin Brodie in 1855 proving graphite was a form of carbon by producing pure graphite from carbon. However, in 1955, American scientist Francis Bundy and coworkers could transform graphite into diamond at high temperature and pressure.
Recent Alotropes of Carbon
A new soccer-ball-shaped carbon allotropes fullerenes were discovered by Robert Curl, Harry Kroto, and Richard Smalley in 1985. The best known form of allotropic fullerene is buckminsterfullerene or C60, consisting of 60 carbon atoms. However, a large number of fullerenes exist with starting from C20 and reaching up to C540.
Graphene is the most recently discovered allotropic form, and it contains a single layer of carbon atoms arranged in a hexagon. The discovery of graphane was announced in 2004 by Kostya Novoselov and Andre Geim. It hosts an indium tin oxide nanoparticle on its surface and generally helps to secure two platinum nanoparticles for improved catalysis in a fuel cell.
Isolation of Carbon
Coke
Among all the allotropic forms, coke is used in the largest quantities and obtained by high-temperature carbonization of coal. It is obtained when coal is heated in a large oven in the absence of air.
Graphite
Natural graphite is obtained as a mixture with mica, quartz, and silicates. It is washed by floatation and heated with HCl and HF in a vacuum when residual silicon compounds are driven away as SiF4.
However, half of the industrial requirement of graphite is fulfilled with synthetic graphite. It is generally obtained by heating silica with coke in an electric furnace (2500 °C) for about 24 hours.
SiO2 + 3C → SiC + 2CO → C (graphite) + Si (g)
Diamond
Natural diamonds are mined in large quantities, more than 18 tonnes a year. Among these, about 30% are used as gems in jewelry, while the rest go to industrial uses.
Only a small amount of industrial demands can be made synthetically by subjecting graphite to 125000 atm pressure at about 3000 K temperature. However, such conversion can be achieved at 70000 atm pressure and 2000 K temperature when transition metal catalysts can be used.
Activated Carbon
The incomplete combustion of hydrocarbons gives us carbon black. However, activated carbon is prepared by controlled pyrolysis of organic materials like sawdust or coconut shell at 900 °C. Activation of the surface is accompanied by adding materials that would oxidize and dehydrate the organic substrate on the surface.
Graphite Fibers
Graphite fibers are obtained when synthetic fibers or asphaltic fibers are subjected to controlled pyrolysis at 1500 °C. The strong fibers have the same structural construction as graphite but constitute a layer of ribbons parallel to the axis of fibers.
The strong bonds in a plane are responsible for high tensile strength. Therefore, these fibers are used to reinforce plastics. Such plastics are used to make tennis rackets, aircraft components, etc.
Properties of Carbon
Carbon is a nonmetal that has 4 electrons in the valence shell. Therefore, it uses 4 electrons to form covalent bonding with other elements, especially hydrogen. The catenation properties of carbon allow for the formation of long carbon chains and rings.
Therefore, it is unique among all the periodic table elements because it has the ability to form strongly bonded chains or rings with hydrogen and itself. The C-C bonds in such chains and rings are nonpolar and stronger.
The element carbon does not melt when heated at normal atmospheric pressure. It does not undergo any liquid phase, but it changes directly from solid to gas. However, when pressure is increased to 10 atmospheres, the allotropic form of graphite is observed to melt at 3550 °C.
| Symbol | C | ||
| Discovery | Such a periodic table element has been known since prehistoric times. | ||
| Origin of the name | The name originates from the Latin word ‘carbo’, charcoal. | ||
| Relative atomic mass | 12.011 | ||
| Atomic number | 6 | ||
| Electron configuration | [He] 2s22p2 | ||
| CAS number | 7440-44-0 | ||
| ChemSpider ID | 4575370 | ||
| Periodic Position | Group 2 period 14 (p-Block) in the periodic table | ||
| Allotropes | Diamond, graphite, graphene, amorphous, fullerene | ||
| Key isotopes | 12C, 13C, 14C | ||
| Appearance at 20°C | Solid | ||
| Melting point | Sublimes at 3825°C, 4098 K | ||
| Boiling point | Sublimes at 3825°C, 4098 K | ||
| Density (g cm−3) | 3.513 for diamond and 2.2 for graphite | ||
| Atomic radius, non-bonded (Å) | 1.70 | ||
| Covalent radius (Å) | 0.75 | ||
| Electron affinity (kJ mol−1) | 121.776 | ||
| Electronegativity | 2.55 (Pauling scale) | ||
| Ionisation energies (kJ mol−1) | 1st | 2nd | 3rd |
| 1086.454 | 2352.631 | 4620.471 | |
| Common oxidation states | 4, 3, 2, 1, 0, -1, – 2, -3, -4 | ||
| Specific heat capacity (J kg−1 K−1) |
709.1 | ||
Chemical Properties
The electronic configuration of C is 1s2 2s2 2p2, and it uses four valence electrons for covalent chemical bonding. Due to the presence of 4 valence electrons, the most common oxidation number or state of carbon is +4. It can also show +2 and 0 oxidation states in various inorganic compounds and carbonyls. A pure element generally forms a huge number of compounds with other elements.
The atomic form of C is very short-lived, but it is stabilised by various allotropic forms. A number of pure forms of carbon are graphite, diamond, fullerenes, and graphene.
All these allotropes contain C atoms in their structure, but the properties of these allotropes are different due to the different structural arrangements of C atoms. Most allotropes of carbon are comparatively unreactive in normal conditions.
Carbon generally participates in the four main types of chemical reactions, such as oxidation, addition, substitution, and combustion.
- Various carbon products are obtained when we heat coal, natural gas, wood, or bone at elevated temperatures in the presence of insufficient oxygen.
- It generally reacts with oxygen to form carbon oxides at elevated temperatures. Such a process is used for the production of various metals from their oxides.
- The fuels made up of carbon and its compounds are burned in air in the presence of oxygen to form heat and light. The process where C and its compounds burn to release energy is called combustion.
Position of Carbon in Periodic Table
The atomic number of carbon is 6, and the electronic configuration of the element is [He] 2s22p2. Therefore, nonmetal carbon is positioned in group 14 and period 2 of the periodic table. The valence shell electronic configuration of carbon suggests that it is a p-block element, which is placed after boron and before nitrogen in the periodic table.
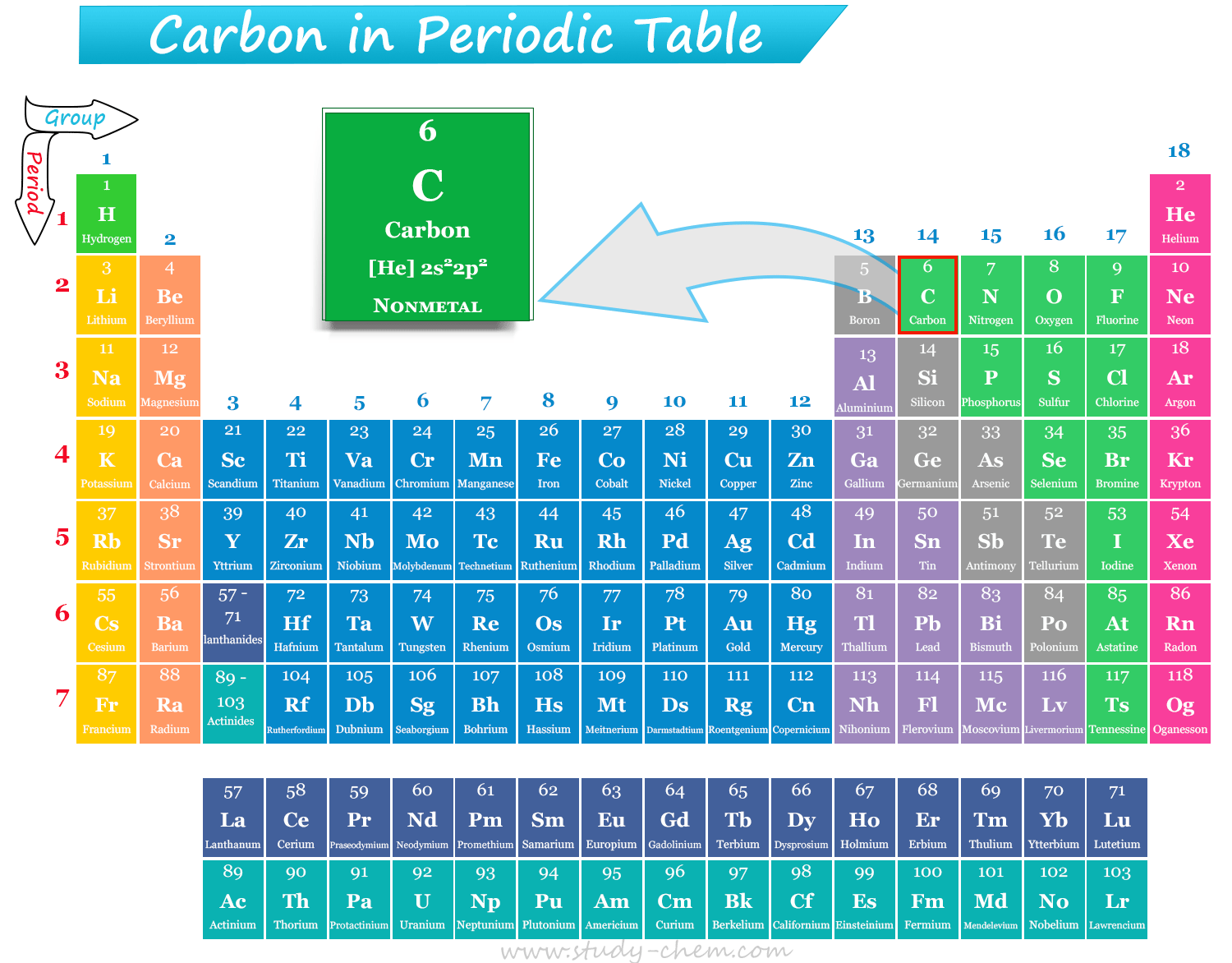
Carbon is the 6th element, or first member of group 14 of the periodic table. Therefore, carbon is placed along with other group 14 elements: Silicon (Si), Germanium (Ge), Tin (Sn), Lead (Pb), and Flerovium (Fl).
Facts of Carbon
- Carbon is a unique chemical element or non-metal in Group 14 of the periodic table that has the symbol C and atomic number 6.
- Catenation is the most amazing properties of carbon, where it make long C chains and rings.
- Formation of pπ-pπ bonds or double or triple bonds is also a special and unique ability of the carbon atom.
- Due to formation catenation and multiple bond formation, it has a number of allotropic forms.
- Carbon atoms contain about 20% of the body weight of living things.
- Carbon is the 4th most abundant element in the universe by mass, and it forms the largest number of chemical compounds among all the periodic table elements.
- The allotrope diamond is the hardest material with the highest thermal conductivity. Therefore, it can grind down any substance by conducting away. heat generated during grinding.
Uses of Carbon
A number of pure and impure forms of carbon are charcoal, coke, graphite, diamond, fullerenes, graphene, carbon black, activated carbon, etc. All these forms of this element have different appearances and different uses.
- Various hydrocarbons are extracted naturally as coal, oil, and natural gas. These are mostly used as a source of fossil fuels.
- A small but important fraction of petroleum products is used in petrochemical industries for producing polymers, fibres, paints, solvents, plastics, etc.
- The impure forms, like charcoal and coke, are used widely in metal smelting. Coke is vital in the extraction of iron and the making of steel.
- Graphite is also an important allotrope of carbon that is used mainly for making steel, metal foundries, refractories, in making cusible, nozzles, etc. It is also used in brake lining, pencils, brushes of electric motors, etc.
- Diamond is a transparent crystalline solid and the hardest and most valuable gemstone that is used widely in jewelry. Industrial diamonds are used for cutting rocks, making drills, and polishing.
- Carbon black is largely used in the rubber industry for making car tyres.
- Activated carbon is a very efficient absorbent that absorbs organic pollutants from drinking water and noxious gases from the air. Therefore, it may be used for the purification of water and air. It is also used as a decolorizing agent in the sugar industry.
- Carbon fibre is a very strong and lightweight material, and it is mostly used in tennis rackets, skis, fishing rods, rockets, and aeroplanes.
- Carbon nanotubes, fullerenes, and atom-thin sheets of graphene have revolutionised the impact on hardware developments.
Importance of Carbon in Human Body
Carbon is the second most abundant element and a basic building block in the human body. It is an essential component for the formation of biomolecules like DNA, proteins, carbohydrates, lipids, etc.
Carbon-based biomolecules (proteins, carbohydrates, lipids) are the main source of energy for the human body.