What is Hydrogen?
Hydrogen, symbol H, molecular formula H2, is a colorless, odorless, tasteless, flammable gaseous chemical substance in the periodic table with atomic number 1. In chemistry or chemical science, the hydrogen atom is the only member of the chemical element in which the valence electron is under the direct influence of the nucleus. Therefore, it bears one unit of positive and negative electrical charge. Hydrogen is an essential element for life, but it does not play an active role in any biological process. However, it is present in all the biomolecules present in living things. Hydrogen gas is a clean fuel for the future because it is a pollution-free source of energy

Hydrogen is a colourless, odourless gas that has the lowest density among all gases. Under ordinary conditions, hydrogen gas generally consists of a pair of atoms or a diatomic molecule with a wide range of bonding. The most important chemical compound, water (H2O), is obtained when burning it with oxygen molecules.
Natural Abundance
Hydrogen is the most abundant element in the universe and is found mostly in the sun and stars. A huge quantity of hydrogen is present on our Earth as water.
Hydrogen is present as a gas in the atmosphere only in tiny amounts because the speed of the H2 molecule at the temperature of atomsphere is sufficient to enable it to escape Earth’s gravitational field. Therefore, any hydrogen that enters the atmosphere can quickly escape the Earth’s gravity into outer space.
Most of this gas, produced during the heating of natural gas with steam, is a mixture of hydrogen and carbon monoxide. Hydrogen gas can also be formed during the electrolysis of water.
History and Discovery
The alchemist Paracelsus noted that bubbles were given when iron filings were added to sulfuric acid. The same observation was also noted by Robert Boyle in 1671.
In 1766, Henry Cavendish collected such bubbles and proved that they were different from other gases. A few years later, he showed that when hydrogen burns, it forms water. The name of the gas was given by Antoine Lavoisier.
Properties of Hydrogen
Due to its unique characteristics and properties, hydrogen (H) forms chemical compounds or molecules with nearly every periodic table element.
| Hydrogen Facts |
|||
| Symbol | H | ||
| Discovery | Henry Cavendish (1766) | ||
| Origin of the name | The name is derived from the Greek words ‘hydro’ and ‘genes’, meaning water forming | ||
| State at 20°C | Gas | ||
| Relative atomic mass | 1.008 | ||
| CAS number | 133-74-0 | ||
| Periodic Properties |
|||
| Atomic number | 1 | ||
| Electron per shell | 1 | ||
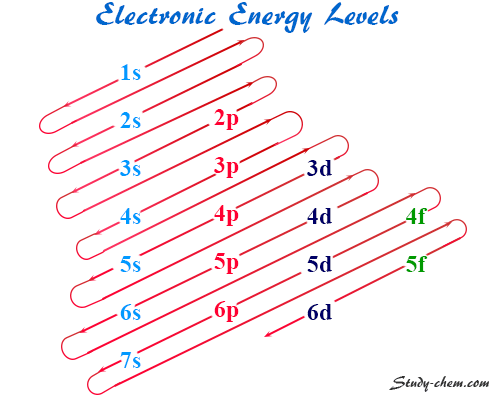
| Electron configuration | 1s1 | ||
| Group | 1 | ||
| Period | 1 | ||
| Block | s-Block | ||
| Physical Properties |
|||
| Melting point | −259.16°C | ||
| Boiling point | −252.879°C | ||
| Density (g cm−3) | 0.000082 | ||
| Atomic properties |
|||
| Oxidation states | −1, +1 | ||
| Atomic radius (Å) | 1.10 | ||
| Covalent radius | 0.32 | ||
| Electron affinity (kJ mol−1) | 72.769 | ||
| Electronegativity (Pauling scale) |
2.20 | ||
| Ionisation energies (kJ mol−1) | 1st | 2nd | 3rd |
| 1312.05 | – | – | |
| Main isotopes | 1H, 2H, 3H | ||
The normal oxidation number or state of hydrogen in chemical compounds is +1, but highly electropositive metals (alkaline and alkaline earth) show a −1 oxidation state.
Hydrogen in Periodic Table
In the modern periodic table, hydrogen is placed in group-1 with alkali metals like lithium, sodium, potassium, rubidium, cesium, and francium.
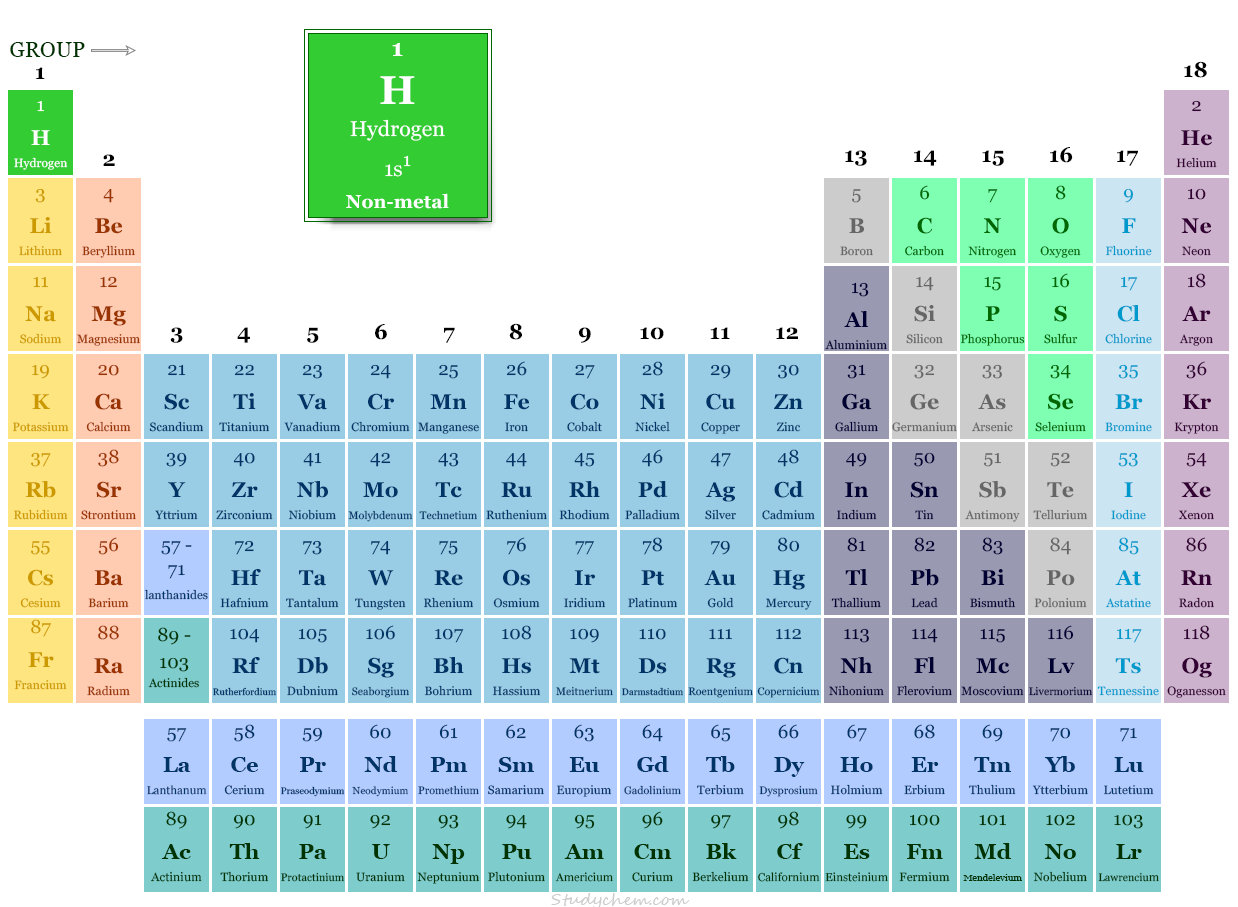
The unique periodic table position can be described by its characteristic properties and electron configuration.
- The ns1 electron configuration justifies its position in group-1 with the alkali metals.
- When considering other facts, the electronic configuration of hydrogen is one electron short of the next noble gas, helium (He). Therefore, it is placed in group 17 in the periodic table with the halogens.
- Due to the presence of a half-filled valence shell, it is also placed in group-16 with the carbon family. Therefore, it forms covalent bonds with a wide range of polarities in chemistry.
Properties of Hydrogen Gas
The chemistry of hydrogen has late gained. It will be used as an alternative source of energy in the near future (fuel cells) due to the huge stock of H2 in the earth’s surface water molecules.
The molecule is a very stable species, at 2000°K and 1-atmosphere pressure, only one percent of atomic hydrogen is present in the gas molecules. Therefore, these facts suggested the lack of reactivity at ordinary temperature and pressure.
During atomic forms, it forms various unstable ionized species like a proton (H+), a hydride ion (H−), and a molecular ion (H2+).
Hydrogen Isotopes
The three isotopes of hydrogen gas molecules are represented as H2 (dihydrogen), D2 (di-deuterium), and T2 (di-tritium). The chemical properties of all these gas molecules are basically the same, but hydrogen isotopes differ in their physical properties. At standard temperature and pressure, H2 gas generally exists as a colorless, odorless, tasteless, highly combustible, non-toxic, nonmetallic chemical compound.
Preparation Formula
Hydrogen is prepared
- By the action of metals on water and acids
- By the electrolysis of water
- The reforming hydrocarbon by the stream.
It is also obtained as a byproduct in the electrolytic manufacture of caustic potash from brine.
Small Scale Preparations
When the alkali and alkaline earth metals like sodium (Na), calcium (Ca), magnesium (Mg), and aluminum (Al) react with water in cold conditions, it evolving hydrogen gas. In hot conditions, metals like Zn, Fe, Co, Ni, and Mn also decompose water.
2Na + 2H2O → 2NaOH + H2
2Al + 6H2O → 2Al(OH)3 + 3H2
Zn + 2H2O → Zn(OH)2 + H2
3Fe + 4H2O → F3O4 + 4H2
Large Scale Production
Very pure H2 and O2 can be obtained during the electrolysis of 20 percent caustic soda or potash solution in steel cells using iron electrodes. The anode is nickel plated to prevent oxidation. The anode and cathode are also separated by a porous diaphragm to prevent diffusion and mixing of the gases.
Large amounts of H2 molecules are now obtained when we reform natural gas (hydrocarbon) by steam in the presence of a nickel catalyst at 900°C.
CnH2n+2 + nH2O → nCO + (2n+1)H2
Some carbon dioxide and methane are also formed by this process. The gas mixture is cooled and removed of carbon dioxide (CO2) and methane(CH4) for the fresh preparation of H2 gas molecules.
Uses of Hydrogen Gas
- H2 is used generally for the manufacture of chemicals like ammonia, hydrochloric acid, alcohols (methanol), aldehyde (formaldehyde), etc. Ammonia is an important component in the production of agricultural fertiliser.
- Cyclohexane and methanol obtained from H2 are used widely in the plastics and pharmaceutical industries
- Hydrogen plays an important role in removing sulfur from fuels during the oil-refining process.
- In the glass industry, it is used for making a protective atmosphere.
- During the manufacture of silicon chips for the electronics industry, it is used as a flushing gas
- The atomic, molecular, and liquid form is also used in fuel cells (hydrogen-oxygen fuel), hydrogenation of oils, welding with atomic torches, reduction of metal ores, and propulsion of rockets (in Saturn-V).
- In bubble chambers, it is used for the study of high-energy particles.
- Due to low density, hydrogen is a natural choice for filling gas balloons.
- Hydrogen has been used recently for the reduction of metal oxides. Therefore, direct reduction of iron ore has now been developed on a commercial basis.
Uses of Liquid Hydrogen
In the near future, liquid hydrogen will be used as an alternative source of energy when our reserves of fossil fuels (coal, oil, and natural gas) are exhausted.
The great advantage of liquid hydrogen is used as fuel. It is free from the pollution hazard of conventional fuels.
The only problems lie with the storage and transmission of large quantities of gases. Therefore, vacuum-isolated cryogenic tanks are used to store huge quantities of liquid hydrogen in the United States, Canada, Germany, and the United Kingdom space programs.