Ionic Bond in Chemistry
Ionic bond or electrovalent bond is defined as the electrostatic forces that bind together oppositely charged ions formed by the transfer of electron or electrons from an electropositive metal atom to the electronegative non-metal atom. Therefore, an ionic bond in an ionic compound is formed between electropositive and electronegative chemical elements by the transfer of electrons from electropositive to electronegative atoms to form cations and anions. Two ions that formed an ionic compound are bound together by the strong electrostatic force of attraction.
Ionic bonds are generally formed when there is a high electronegativity difference between the two atoms of the chemical elements. The alkali and alkaline earth metals are highly electropositive and have very low electronegativity. Similarly, group 17 and group 16 non-metals are highly electronegative and have very high electronegativity. Such electronegativity is responsible for ionic bonding between these two types of elements.
The anion formed during ionic bonding always achieves an inert gas configuration, while the cation formed may achieve any of the following configurations:
- Inert gas configuration
- Pseudo-inert gas configuration
- The inert pair configuration
- ns2p6dz type configuration
- Irregular configurations
Formation of Ionic Bond
The atoms of the elements that have almost full or almost empty electrons in their valence shell are very reactive. For example, atoms of alkali (group 1) and alkaline earth metals (group 2) have one or two electrons in their valence shell. Therefore, atoms of group 1 and group 2 can easily transfer such electrons to almost empty non-metal atoms of group 17 and group 16 and combine together by ionic bonding.
The high difference in electronegativities between these two types of elements favours the formation of ionic compounds between them. When the atoms of these two types of elements transfer electrons, they achieve a stable, inert gas electronic configuration.
- The lower the charge on cations, the better the chance for the formation of ionic compounds. Since the lower charge on the cations corresponds to low ionization energy. The larger size of the metal atom or ion favors the formation of ionic compounds. Since an increase in size is associated with a decrease in ionization energy.
- Small size and low charge on the anions will favor the formation of ionic compounds. These factors are also associated with increases in electron affinity and electronegativity.
Such a conclusion predicts which elements are most likely to form ionic compounds. Thus, the elements that have low ionization energy are the best candidates for forming positive ions. Similarly, the elements that have high electron affinity are most likely to form negative ions.
The above discussion has given us a rough guide for the polarity of ionic compounds. But unexpected result sometimes observed. The second electron affinity of oxygen is negative, but it forms a stable ionic compound with alkali and alkaline earth metals.
Ionic Bond and Compounds
Chemical compounds that are formed when cations and anions are held together by electrostatic lines of force are called ionic compounds. The cation and anion that hold together are generally a metal cation and a non-metal anion. NaCl, MgO, KCl, CaO, CaCl2, MgCl2, K2O, etc are the common examples of ionic compounds.
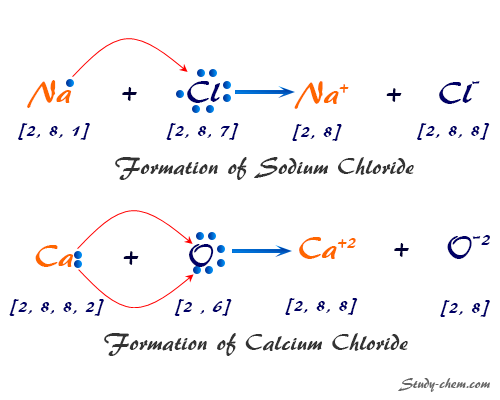
Formation of Sodium Chloride (NaCl)
Let us consider the ionic bonding between a sodium and a chlorine atom.
The electronic configuration of sodium: 1s22s22p63s1 or [Ne] 3s1
The electronic configuration of chlorine: 1s22s22p63s23p5 or [Ne] 3s2 3p5
- A sodium atom loses an electron by absorbing energy equal to its ionization energy and is converted into a cation, Na+. After losing one valence electron, the electronic configuration of the sodium ion (Na+) is 1s22s22p6.
- The chlorine atom picks up the electron released by the sodium atom and is converted into an anion, Cl−. After gaining one valence electron, the electronic configuration of the chloride ion (Cl−) is 1s22s22p6. In this process enrgy equal to electron affinity is released.
- The cation (Na+) and anion (Cl−) combine together by the electrostatic force of attraction to form a stable ionic crystal of sodium chloride (NaCl).
An examination shows that the electronic configuration of Na+ and Cl− ions is a stable noble gas configuration. Therefore, the attainment of a stable noble gas configuration is the clue to this chemical bond.
Formation of Magnesium Oxide (MgO)
The electronic configuration of magnesium and oxygen is:
Magnesium (atomic number 12): 1s2 2s2 2p6 3s2 [Ne] 3s2
Oxygen (atomic number 8): 1s2 2s2 2p4 or [He] 2s2 2p4
From the above electronic configuration, magnesium has two electrons in its valence shell, and oxygen has six electrons in its valence shell.
- A magnesium atom easily loses two valence electrons by absorbing energy to convert into a cation, Mg2+. After losing two valence electrons, the magnesium ion (Mg2+) gets a stable noble gas configuration of 1s2 2s2 2p6 or neon.
- The oxygen atom picks up these two electrons released from the magnesium atom to form an anion, O2−. After gaining two valence electrons, the oxygen ion (O2−) also gets a stable noble gas configuration of 1s2 2s2 2p6 or neon.
- The cation (Mg2+) and anion (O2−) combine together by the electrostatic force of attraction to form a stable ionic crystal of magnesium oxide (MgO).
Formation of Calcium Chloride (CaCl2)
The electronic configuration of calcium and chlorine is:
Calcium (atomic number 20): 1s2 2s2 2p6 3s2 3p6 4s2 or [Ar] 4s2
Chlorine (atomic number 17): 1s2 2s2 2p6 3s2 3p5 or [Ne] 3s2 3p5
From the above electronic configuration, calcium has two electrons in its valence shell, and chlorine has seven electrons in its valence shell.
- A calcium atom easily loses two valence electrons by absorbing energy to convert into a cation, Ca2+. After losing two valence electrons, the magnesium ion (Ca2+) gets a stable noble gas configuration of 1s2 2s2 2p6 3s2 3p6 or argon.
- Two chlorine atoms pick up these two electrons released from the calcium atom to form two anions, 2Cl−. After gaining one valence electron, each of the chloride ions (Cl−) also gets a stable noble gas configuration of 1s2 2s2 2p6 3s2 3p6 or argon.
- The cation (Ca2+) and two anions (2Cl−) combine together by the electrostatic force of attraction to form a stable ionic crystal of calcium chloride (CaCl2).
Formation of Potassium Oxide (K2O)
The electronic configuration of calcium and chlorine is:
Potassium (atomic number 19): 1s2 2s2 2p6 3s2 3p6 4s1 or [Ar] 4s1
Oxygen (atomic number 8): 1s2 2s2 2p4 or [He] 2s2 2p4
From the above electronic configuration, potassium has one electron in its valence shell, and oxygen has six electrons in its valence shell.
- Two potassium atoms easily lose one electron from each by absorbing energy to form two cations, 2K+. After losing one valence electron, each potassium ion (K+) gets a stable noble gas configuration of 1s2 2s2 2p6 3s2 3p6 or argon.
- An oxygen atom picks up these two electrons released from two potassium atoms to form an anion, O2−. After gaining two valence electrons, the oxygen ion (O2−) also gets a stable noble gas configuration of 1s2 2s2 2p6 or neon.
- Two potassium ions (2K+) and one oxide ion (O−2) combine together by the electrostatic force of attraction to form a stable ionic crystal of potassium oxide (K2O).
Characteristics of Ionic Compounds
- The cations and anions in ionic compounds are held together by strong electrostatic forces of attraction. A considerable amount of energy is required to break the ionic compound. Therefore, ionic compounds are hard and stable.
- Ionic compounds are polar and generally soluble in polar solvents (water, ammonia, etc). However, they are insoluble in non-polar solvents (carbon tetrachloride, benzene, etc). It loses its crystal lattice when dissolved in water or any other polar solvent.
- Such compounds are easily ionizable in solution or in the fused state.
- Ionic solids are poor conductors of electricity because the positive and negative ions present in the ionic compound are fixed in their positions. Therefore, such ions cannot move even if an external electric field is applied to them.
- In fused state, ionic compounds conduct electricity because the positive and negative ions in fused state become free to move under the applied electric field.
- The solutions of ionic compounds are good conductors of electricity because ionic compounds give free ions in solution.
- They possess high melting and boiling points due to the formation of a crystal lattice.
- The polar linkages present in ionic compounds are non-directional, and they commonly form a giant type molecule. Each ion is surrounded by the greatest possible number of oppositely charged ions. Therefore, no separate or discrete molecules are present in the crystal lattice of an ionic compound.