Beryllium in Periodic Table
Beryllium is an alkaline earth metal or group 2 periodic table element that has the symbol Be and atomic number 4. It is a lightweight, steel-gray, relatively soft alkaline earth metal that is used for alloying with copper or nickel to make gyroscopes, springs, electrical contacts, spot-welding electrodes, and non-sparking tools. Such a brettle, steel-gray solid metal has 4 protons in its nucleus. The name of the metal beryllium originates from the Greek name of the gemstone beryl, “beryllo.” It was discovered in 1797 by French chemist Nicholas Louis Vauquelin.

Beryllium and its compounds do not play any biological role in human beings or other living systems. A small part of Be, about 0.4 ppb (parts per billion), is found in the human body, but it is carcinogenic and too toxic.
Due to its high reactivity, the free form of beryllium does not occur in nature. Many beryllium-containing minerals are found in our earth crust, but not all of these minerals contain an extractable amount of such alkali metal. The most common beryllium-containing mineral that is used for the extraction of said metal is beryl (Al2Be3Si6O18), bertrandite (Be4Si2O7(OH)2), phenakite (Be2SiO4), and chrysoberyl (BeAl2O4).
Abundance and Occurrence
Beryllium has a very low abundance in the Earth’s crust. However, a very small quantity of Be metal is found in 30 different minerals on Earth. Among these, the most important and accessible surface deposits are beryl (beryllium aluminum silicate) and bertrandite (beryllium silicate).
Emerald and aquamarine are the precious forms of the mineral beryl. They are the beautiful crystals that are often used as gems. The green or bluish green color of the crystalline emerald is due to the presence of a small amount of chromium (III) ion.
The last portion of crytalization of pegmatite rock contains a small amount of Be metal. Due to its small size, beryllium can replace Si in the SiO4 unit. However, the charge balance is inappropriate between Be (II) and Si (IV). It is one of the several reasons for the late crystallization of Be in pegmatite rock.
Most of the Be in the world is mined in Utah, USA. Therefore, the USA is the major producer that generates more than 87% of the Be used in the world. Such an alkaline earth metal is generally obtained by reducing beryllium fluoride with metallic magnesium.
Discovery of Be
The alkaline earth metal beryllium was discovered in 1797 by French chemist Nicholas Louis Vauquelin. He discovered this new element when analyzing beryl and emerald on a suggestion from French mineralogist Abbé René-Just Haüy. Vauquelin announced his discovery in February 1798 at the French Academy in the journal Annales de chimie et de physique.
In this journal, he named it glaucinium (derived from the Greek “glykys” meaning sweet) because of the sweet taste of some of its compounds. The present name beryllium was first used by Friedrich Wöhler in 1828. However, both the names beryllium and glucinum were used concurrently until 1949.
Such an alkaline earth metal was extracted independently in 1828 in Berlin and Paris by two chemists, Friedrich Wöhler and Antoine-Alexandere-Brutus Bussy. They extracted it from beryllium chloride by reacting it with potassium.
BeCl2 + 2 K → 2 KCl + Be
However, Paul Lebeau in 1898 extracted pure (99.5 to 99.8%) beryllium metal during the direct electrolysis of a molten mixture of beryllium fluoride and sodium fluoride.
Extraction Process
Beryllium is relatively rare in our universe, and most people not seen the pure form of this metal. Most of this alkaline earth metal is extracted in three countries (the United States, China, and Kazakhstan) from beryl. The most common extraction process of pure beryllium metal from Beryl (B3Al2Si6O18) is:
- Beryl (B3Al2Si6O18) is roasted with NaF and Na2SiF6 at 700 to 750 °C to form insoluble sodium hexafluoroaluminate, Na3AlF6 and soluble sodium tetrafluoroberyllate, Na2BeF4.
- The roasted mass is leached with boiling water to separate sodium tetrafluoroberyllate (Na2BeF4).
- When sodium hydroxide (NaOH) is added to the extract, a precipitate of beryllium hydroxide, Be(OH)2 is formed.
- We also added ammonium hydrogen fluoride, NH4HF2 to convert Be(OH)2 to ammonium fluoberyllate, (NH4)2BeF4.
- When ammonium fluoberyllate is heated at 900 °C, it forms BeF2.
- BeF2 is reduced by magnesium at 1300 °C to form beryllium metal.
The last step proceeds rapidly at 900 °C. However, the temperature is raised to 1300 °C (above the melting point of Be) for the easy separation of MgF2.
Position of Beryllium in Periodic Table
Beryllium is a silvery white lustrous alkaline earth metal that is positioned in group 2 and period 2 of the periodic table. The atomic number of beryllium is 4, and the electronic configuration of the metal is [He] 2s2. Therefore, beryllium is an s-block element that is placed after lithum and before boron in the periodic table.
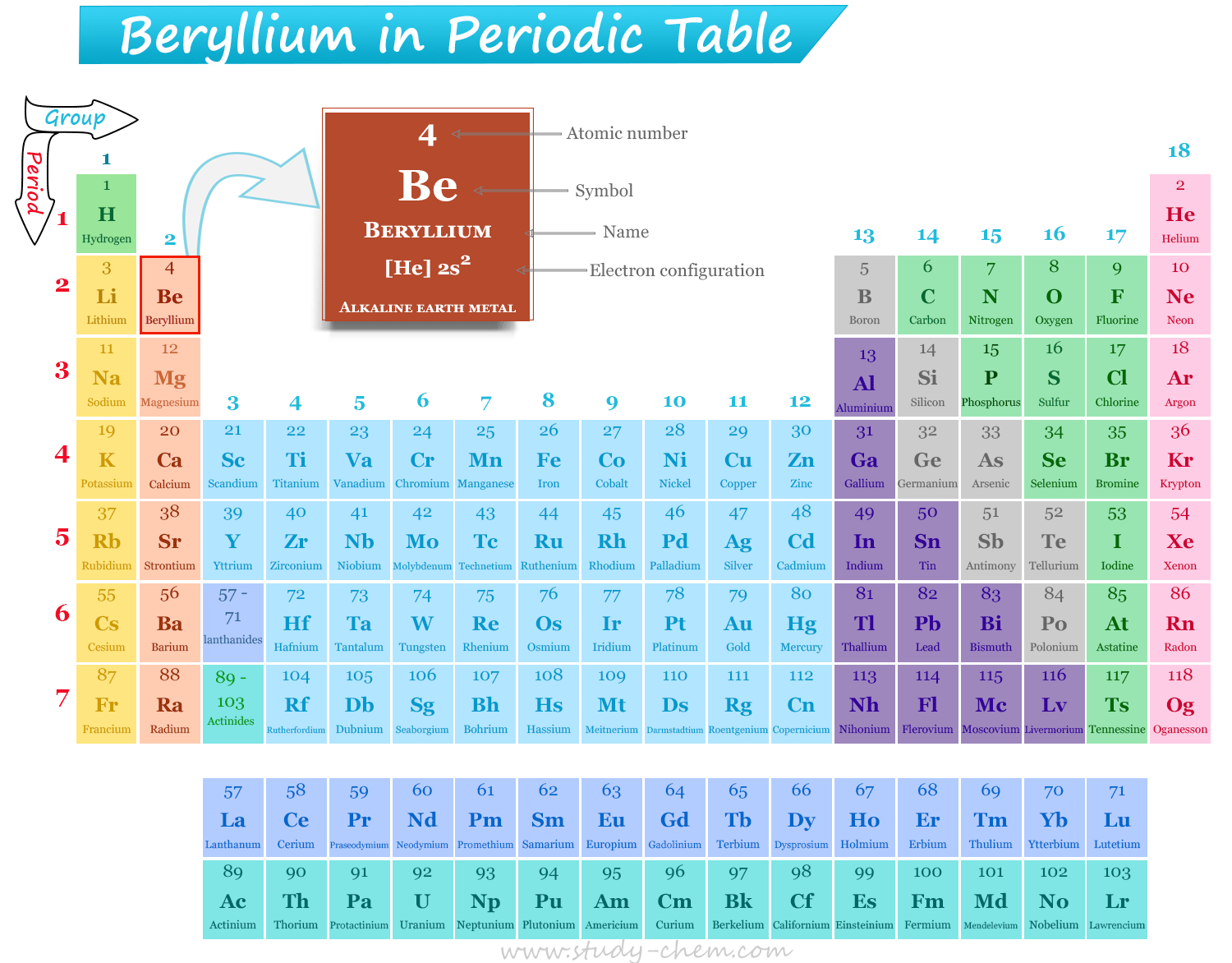
It is the 4th element, or located at the top of the second column or group of the periodic table. Therefore, beryllium is placed along with other alkaline earth metals (magnesium, calcium, and strontium) in the periodic table.
Properties of Beryllium
Beryllium is less reactive than other alkaline earth metals because it forms an oxide layer on the reactive surface at normal temperatures. However, such a layer can break when the temperature rises and react with air, acids, bases, halogens, etc.
The powdered form of the metal burns easily in air to form metal oxide and nitride because such a form is a more reactive form of Be.
Due to its small size and high ionization energy, Be does not react with water and hydrogen. It make Be unique from the other members of such alkaline earth metal group.
| Symbol | Be | ||
| Discovered by | Nicholas Louis Vauquelin in 1797 | ||
| Origin of the name | The name of the element originates from the Greek name for beryl, ‘beryllo’ | ||
| Color/appearance | Silvery-white Luster | ||
| State at 25 °C | Solid | ||
| Relative atomic mass | 9.012 | ||
| Atomic number | 4 | ||
| Electron configuration | [He] 2s2 | ||
| Periodic position | Group 2 and Period 2 (s-block) in the periodic table | ||
| Melting point | 1287 °C | ||
| Boiling point | 2468 °C | ||
| Density (g cm−3) | 1.85 | ||
| State at 20°C | Solid | ||
| Key isotopes | 9Be | ||
| Atomic radius, non-bonded (Å) | 1.53 | ||
| Covalent radius (Å) | 0.99 | ||
| Electron affinity (kJ mol−1) | Not stable | ||
| Electronegativity (Pauling scale) | 1.57 | ||
| Ionisation energies (kJ mol−1) | 1st | 2nd | 3rd |
| 899.504 | 1757.108 | 14848.767 | |
| Common oxidation states | 2 | ||
| CAS Number | 7440-41-7 | ||
Chemical Properties
The element Be has two valence electrons in an s-shell outside an inert gas core. These two electrons are always involved together to give rise to the uniform valency of the element. Therefore, two electrons (per atom) are available for chemical bonding.
The high value of thired ionization energy is quite expected for the closed shell electronic configuration of the Be+2 ion. It explains the upper limit of the +2 oxidation state of the element.
Similarities between Beryllium and Aluminum
The charge by radius ratio for Be+2 is similar to that of the Al+3 ion. It suggests that Be and Al have very similar chemical properties. The common similarities of these two elements are:
- Both metals dissolve in aqueous alkali, evolving hydrogen.
- The standard electrode potential of these two metals is equal.
- The oxides and hydroxides of these two metals are amphoteric.
- The anhydrous halides of both metals are dimeric with metal-halogen-metal bridges.
- The carbides of the metals yield methane when dissolved in water.
- Both metals, Be and Al, form a protective oxide layer on the surface.
- Both are rendered passive by concentrated nitric acid.
- The thermal stability of the anhydrous sulfates is nearly the same.
Uses of the Beryllium Element
- Beryllium is alloyed with copper or nickel to increase their strength, thermal, and electrical conductivity. Therefore, such alloys are good to make gyroscopes, springs, electrical contacts, spot-welding electrodes, and non-sparking tools.
- Many other Be alloys are used for making high-speed aircraft, missiles, spacecraft, and communication satellites.
- The oxides of beryllium have very high melting points and are excellent thermal conductors and electrical insulators. Therefore, they are very useful for nuclear work and ceramic applications.
- In nuclear reactors, beryllium is used as a reflector or moderator of neutrons.
- Due to its low atomic weight and low absorption of X-rays, it is used as a window material in X-ray tubes.
- In military industries, beryllium foil may be used for making nuclear weapons.
Frequently Asked Questions
What is Beryllium?
Beryllium is a lightweight, steel-gray, soft alkaline earth metal that is positioned in group 2 and period 2 of the periodic table. It is alloyed with copper or nickel to make gyroscopes, springs, electrical contacts, spot-welding electrodes, and non-sparking tools.
Who discovered beryllium?
The alkaline earth metal beryllium was discovered in 1797 by French chemist Nicholas Louis Vauquelin.
Is Beryllium Dangerous for Human Beings?
Yes, beryllium is dangerous for human beings.
The solid metal is slightly safe for human health. However, when we inhale beryllium fumes and dust, it may cause chronic inflammation in the lungs. Therefore, such inhalation can increses the risk of lung cancer.
Where was beryllium discovered?
The metal beryllium was discovered in 1798 in France. Vauquelin announced his discovery in February 1798 at the French Academy in the journal Annales de chimie et de physique.
What is beryllium used for?
When a small amount of beryllium is added to copper, it forms an alloy six times stronger than pure copper metal. It is largely used to improve the strength of copper and nickel.
Such alloys are non-magnetic, corrosion-resistant, and possess high strength and good electrical conductivity. Therefore, they are extensively used for making critical moving parts of aero engines and many other instruments like gyroscopes, springs, electrical contacts, spot-welding electrodes, and non-sparking tools.
The element is critically important for electronics because it is a p-type dopant in semiconductor technology. Therefore, it is widely used in the electrical industries for making cell phones, cameras, and in the fine-tuning knobs of radios, thermostats, and lasers.
How many neutrons does beryllium have?
Such a brettle, steel-gray solid metal has 4 protons in its nucleus. A stable beryllium atom has 4 neutrons in its nucleus and 4 electrons in its electronic shell.
Does beryllium react with water?
Due to its small size and high ionization energy, beryllium does not react with water or steam. It make Be unique from the other members of such alkaline earth metal group.
Does beryllium burn in air?
Beryllium is less reactive than other alkaline earth metals because it forms an oxide layer on the reactive surface at normal temperatures. However, such a layer can break when the temperature rises and react with the air.
The powdered form of the metal burns easily in air to form metal oxide and nitride because such a form is a more reactive form of Be.