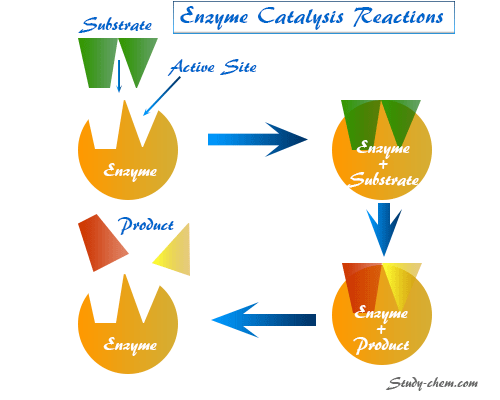
Enzymes Meaning in Science
Enzymes are bioproteins that act as a catalyst in living organisms and regulate the rate of biochemical reactions without being altered themselves. In science, most of the biochemical reactions in living organism regulates by enzymes. Therefore, without enzymes, many of the biochemical reactions would not take place in living organisms. However, an enzyme can catalyze only one type of biochemical reaction and interact with only one type of substance or group of substances called the substrate. When an enzyme is isolated from the cells, they retain their catalytic activity. Except ribosolmes, all enzymes are proteins, and they are generally composed of one or more amino acid chains or polypeptide chains. A deficiency of a particular enzyme may cause many inherited diseases, such as albinism and phenylketonuria.
Enzymes catalyze the reactions during the digestion of food, where large nutrient molecules such as proteins, carbohydrates, and fats are broken down. Therefore, such biomolecules can be broken down into smaller molecules by the conversion or transformation of chemical energy. They are also used in medicines, including killing disease-causing microorganisms, promoting wound healing, and diagnosing certain diseases.
Enzymes are produced from the leaving plant and animals from our environment, and enzymolysis reactions are examples of kinetics reactions where enzymes act as a catalyst. These are responsible for catalyzing an infinite no of chemical changes occurring in the living cells.
Structure of Enzymes
Enzymes are colloidal in nature and three-dimensional complex protein molecules in science with specific characteristics for the formation of enzyme-catalyzed reactions. In chemistry, enzymes disperse in water and form a colloidal state having dimensions in the colloidal range (10-7 to 10-4 cm).
The sequence of amino acids determines the specificity of enzyme molecules. During the fluctuations in temperature or pH, the enzyme can denature or lose its integrity and enzymatic ability. However, denaturation is sometimes reversible but not always.
Cofactor
Some enzymes can bind to an additional chemical component called a cofactor. It can directly participate in the catalytic event and is required for enzymatic activity. A cofactor may be an organic molecule, such as a vitamin, or an inorganic metal ion, or some enzymes require both.
Nomenclature and Classification of Enzymes
In the early days, the names of the enzymes were given by their discoverers in an arbitrary manner. However, most of the names have been obtained by adding the suffix “-ase” to the substrate’s name.
For example, urease acts on urea, and it catalyzes the breakdown of urea. However, lipase acts on lipids, nuclease on nucleic acids, and lactase on lactose. These are trivial names, but not all enzymes are named in this manner. Therefore, a basic classification system has been developed for the nomenclature of enzymes.
Since 1964, the International Union of Biochemistry can clasify enzyme into six major classes based on the type of reaction the enzyme catalyzes. These six principal categories and their biochemical reactions are
- Oxidoreductases
- Transferases
- Hydrolases
- Lyases
- Isomerases
- Ligases
Oxidoreductases
Oxidoreductases are involved in oxidation-reduction or electron transfer reactions. For example, alcohol dehydrogenase, cytochrome oxidase, etc.
Transferases
Transferases are the enzymes that catalyze the transfer of functional groups, such as phosphoryl, glycosyl, and methyl from one substance to another. For example, hexokinase, transaminases, transmethylases, and phosphorylase.
Hydrolases
Hydrolases are enzymes that bring about the hydrolysis of various compounds by the addition of water. For example, lipase, choline esterase, acid and alkaline phosphatases, pepsin, and urease.
Lyases
Lyases are specialized for forming double bonds by adding or removing a chemical group. Aldolase, fumarase, and histidase are examples of such a class of enzymes.
Isomerases
Isomerases are enzymes that are involved in all the isomerization reactions by transferring a group within a molecule to form an isomer. Triose phosphate isomerase, retional isomerase, and phosphohexose isomerase are examples of this class of enzymes.
Ligases
Ligases catalyze the synthetic reactions where two molecules are joined together, and adenosine triphosphate (ATP) or a similar nucleotide is used. For example, Glutamine synthase, acetyl-CoA carboxylase, succinate thiokinase.
Enzyme Kinetics
The kinetic behavior of enzymes is similar to that of the heterogeneously catalyzed reaction. They provide various functional groups at the catalytic site that can become in equilibrium with the substrate molecule and thereby catalyze a chemical reaction. Therefore, the enzyme-catalyzed reactions have often been referred to as micro-heterogeneous zero-order kinetic reactions.
Examples of Enzyme Catalysis Reactions
Enzyme-catalyzed reactions are highly specific in nature. For example, the urase can catalyze the hydrolysis of urea, but it has no effect on the hydrolysis of methyl urea.
Diastase converts starch to maltose, and maltase converts it to glucose. This is on further action with the enzyme zymase to form alcohol.
Specific Characteristics of Enzymes
The enzyme is the copolymers of amino acids with specific amino acid sequences. The enzyme is poisoned and activated like other heterogeneous catalysts.
The activity of enzymes increases experimentally with the rise in temperature, but when the temperature is significantly raised, they lose their activity as they coagulate. The activity formed a maximum with a temperature rise of 35 to 45 centigrade.
Kinetics of Enzymolysis Reaction
The enzymes are also very effective catalysts in the different laboratory processes. The concentration of the enzyme required is very much lower than that of the substrate. The rate of enzymolysis is found to be directly proportional to the concentration of enzymes.
When the concentration of substrate varied, enzymolysis reactions usually formed the initial first-order chemical kinetics at low substrate concentration and approached zero-order kinetics when substrate concentration increased.
Applications of Enzymes
Enzymes are useful therapeutic agents and analytical reagents. They are also used in genetic manipulation in genetic engineering.
Therapeutic Agents
Some enzymes are useful therapeutic agents and has a useful medical applications.
- Streptokinase and urokinase are useful to remove the blood clots.
- The enzyme asparaginase is used in the treatment of tumor cells in leukemias.
- Papain is an anti-inflammatory agent that is used to reduce swelling, pain, and inflammation.
- Pancreatic enzymes (trypsin and lipase is used in pancreatic diseases.
- Certain recombinant enzymes (β-glucosidage, α-glucosidage and α-galactosidage A) have been approved for the treatment of some lysosomal storage diseases.
Analytical Reagents
Some enzymes are useful analytical reagents in the clinical laboratory for the measurement of substrates, durgs or even the activities of other enzymes. Using such specific enzymes gives more accurate results than conventional chemical compounds.
The biochemical compounds, glucose, urea, uric acid, and cholesterol can be more accurately and specifically estimated by enzymes glucose oxidase and peroxidase, urease, uricase, and cholesterol oxidase.
Enzymes in Genetic Manipulation
Many enzymes are used in genetic engineering for manupulation of genes. For example, restriction endonucleases are used for gene transfer and DNA fingerprinting. Another synthetic enzyme, Taq DNA polymerase, is used in polymease chain reaction.
Industrial Applications of Enzymes
Enzymes are valuable biomolecules that have many industrial applications, such as fermenting of wine, leavening of bread, curdling of cheese, brewing of beer, etc.
- Renin is used in the curdling of cheese
- Glucose isomerase is used in the production of high fructose syrup
- α Amylase, β amylase, xylanases, and lipases are used in the food industry to improve texture, enhance flavor, and accelerate fermentation of wine, and in the brewing of beer.
- The enzyme, protease is used for making washing powder
Frequently Asked Questions
What are enzymes?
Enzymes are biocatalysts or three-dimensional complex protein molecules that have been synthesized in living cells and increase the velocity or rate of a biochemical reaction without themselves undergoing any change in the overall process. Except for the ribosome, all the enzymes are proteins in nature. The names of most of the enzymes are given by adding the suffix “-ase” to the substrate’s name.
Which enzyme is not a protein in nature?
Ribosomes are a group of ribonucleic acids that function as biocatalysts, and they are regarded as non-protein enzymes.
Why are enzymes important in industrial processes?
Enzymes are important biomolecules that not only play a crucial role in biochemical reaction but they also have many industrial applications. For example, renin is used in the curdling of cheese and glucose isomerase is used in the production of high fructose syrup.
Many other enzymes, such as α Amylase, β amylase, xylanases, and lipases are used in the food industry to improve texture, enhance flavor, and accelerate fermentation of wine, and in the brewing of beer.
Name two enzymes that are used in genetic engineering.
The enzyme, restriction endonuclease, is used for gene transfer and DNA fingerprinting. Another synthetic enzyme, Taq DNA polymerase, is used in polymease chain reaction.