What is Quantum Number?
Quantum number or principal, azimuthal, magnetic, and spin quantum numbers in chemistry or physics describe the size, shape, and orientation of an electron in an atom. Therefore, the quantum numbers are the identification numbers for an individual electron of an atom which describe the position and energy of an electron in an atom. These quantum numbers are designated generally by the symbol n for the principal quantum number, l for the azimuthal quantum number or orbital angular quantum number, and m or ml for the magnetic quantum number.
Bohr’s model could not explain the fine structure of an atom because the appearance of several spectral lines by only one quantum number suggested by Bohr is not sufficient. The fact clearly indicates that many other quantum numbers are needed to explain the various spectra of an atom.
Principal Quantum Number
The principal quantum number defines the size and energy level, or principal shell to which an electron can stay. It is generally denoted by the letter n.
For the hydrogen atom, the energy of an electron is fixed due to the fixed value of n. However, in other multielectron atoms, the energy of each electron depends mostly on the principal quantum number of the electron. When the value of n increses, the radius or nucleus-electron separation increases. Therefore, the size of the orbital increses and the energy also rises.
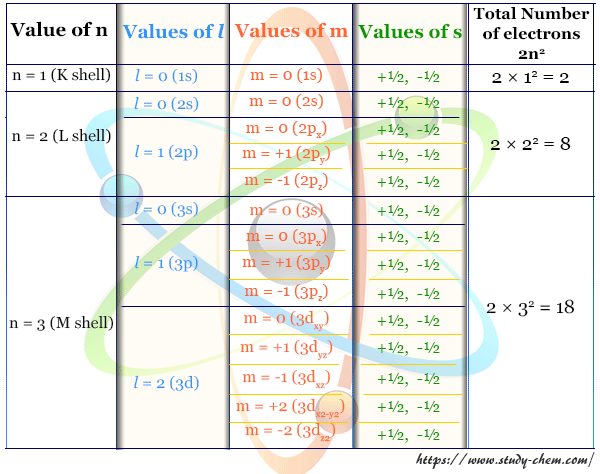
The maximum number of electrons in the principal shell is equal to 2n2, where n = principal quantum number.
| Principal Quantum Shell | Principal Quantum Number | Maximum Number of Electrons |
| K shell | 1 | 2×12 = 2 |
| L Shell | 2 | 2×22 = 8 |
| M shell | 3 | 2×32 = 18 |
| N shell | 4 | 2×42 = 32 |
| O shell | 5 | 2×52 = 50 |
The maximum number of electrons by 2n2 formula can be achieved only for the first four quantum shells (K, L, M, and N). However, no known element among the 118 periodic table elements has more than 32 electrons in any one quantum shell.
The subshells are filled according to the Aufbau principle and to achieve more than 32 electrons in one shell, it needs to go into the g orbital. Presently, we can not know any periodic table element which use g orbital for its electronic arrangement.
How to find the principal quantum number?
The principal quantum number (n) is always an integer, and it can only have a value from 1 to ∞, but 1 to 7 can be established. The capital letters K, L, M, and N are also used to designate the values of n.
If for an electron n = 3, it resides in the M-shell. Therefore, it gives the idea of the size of the shell.
From this idea, we can easily find out the principal quantum shell of an electron. The principal quantum shell of the 6th electron of the calcium atom can be find out following way.
The atomic number of calcium = 20, and it has 20 electrons distributed in different quantum shells. The K shell of the calcium atom has 2 electrons, the M shell has 8 electrons, and the L shell has 12 electrons. Therefore, the 6th electron of the calcium atom resides in the M-shell and the quantum number of the M shell = 2.
The electronic configuration of an atom is also used to find out the principal quantum numbers of an electron in an atom. For example, the electronic configuration of a calcium atom = 1s2 2s2 2p6 3s2 3p6 4s2. Therefore, the 6th electron of the calcium atom resides in the 2p orbital with principal quantum number = 2.
Azimuthal Quantum Numbers
Azimuthal or angular momentum or orbital quantum number is denoted by the letter l. It was introduced by Somerfield in his atomic model to describe the general geometric shape and also the fine structure of the electron wave (orbital).
The azimuthal quantum numbers (l) can have any values from 0 to (n−1) for a given value of n. Therefore, l = 0, 1, 2, … (n−2), (n−1).
How to find the azimuthal quantum number?
The total number of different values of l is equal to n. Different l values define different types of orbitals like s (l=0), p (l = 1), d (l = 2), f (l = 3), and g (l =4). Such quantum numbers and orbital designation can be described in the following manner:
| Principal Quantum Number (n) and Main Shell Designation | Azimuthal Quantum Number (l) and Subshell Designation | Maximum Number of Electrons in different Sub-shells = 2(2l+1) | Total Number of Electrons in the Main Shell (2n2) |
| n = 1 (K-shell) | l = 0 (1s) | 2 | 2×12 = 2 |
| n = 2 (L-shell) | l = 0 (2s) | 2 | 2×22 = 8 |
| l = 1 (2p) | 6 | ||
| n = 3 (M-shell) | l = 0 (3s) | 2 | 2×32 = 18 |
| l = 1 (3p) | 6 | ||
| l = 2 (3d) | 10 | ||
| n = 4 (N-shell) | l = 0 (4s) | 2 | 2×42 = 18 |
| l = 1 (4p) | 6 | ||
| l = 2 (4d) | 10 | ||
| l = 3 (4f) | 14 | ||
| n = 5 (O-shell) | l = 0 (5s) | 2 | 2×52 = 50 |
| l = 1 (5p) | 6 | ||
| l = 2 (5d) | 10 | ||
| l = 3 (5f) | 14 | ||
| l = 4 (5g) | 18 |
In 1s designation, the number 1 stands for the value of n and the latter s denotes l = 0. Similarly, for 3d designation, the number 3 stands for the value of n and the latter d denotes l = 2.
Azimuthal quantum number of an electron
From the electronic configuration data, we can easily find out the azimuthal quantum numbers of an electron in an atom. The electronic configuration of magnesium (atomic number 12) is 1s2 2s2 2p6 3s2. Therefore, the last electron in the magnesium atom resides in the 3s orbital with azimuthal quantum number = 0.
Similarly, the 10th electron of the magnesium atom resides in the 2p orbital with principal and azimuthal quantum numbers = 2, 1.
Magnetic quantum number
We already see that the Bohr Model could not explain the fine structure or splitting of a single spectral line into a number of closely spaced spectral lines in the presence of a magnetic or electric field. The presence of more spectral lines in a magnetic field or in an electric field suggests that the energy levels are further subdivided. To explain such phenomena, Linde introduced a third quantum number called the magnetic quantum number.
The third quantum number m defines the orientation of an electron wave or orbital with respect to a given direction in the presence of a strong electric or magnetic field. It does not affect the shape of the orbital or the energy of an electron, but is used to specify the sub-level of an atom.
How to find the magnetic quantum number?
For a given value of l, m has any integral value between −l to +l, including 0. Therefore, m = −l, ..,−2, −1, 0, … (l−2), (l−1), +l ; where the total number of values of m equals (2l +1).
- For the s wave, l = 0 and m also has 0. Therefore, the s-orbital has only one orientation in space.
- For the p wave, l = 1, and m has the values −1, 0, and +1. These three orientations are expressed by the subscripts px, py, and pz.
- d-type electron wave, l = 2 and m = −2, −1, 0, +1, +2. Thus, five m values describe the five orientations of space like dxy, dyz, dxz, dx2-y2, and dz2.
- For f wave, l = 3 and m = −3, −2, −1, 0, +1, +2, +3. Thus, seven m values describe the seven orientations of space, fy(3x2−y2), fz(x2−y2), fyz2, fz3, fxz2, fxyz, fx(x2−3y2).
The orbital designation and quantum numbers are described in the following table.
| Azimuthal Quantum Number and Orbital Designation | Magnetic quantum number | Total Number of electrons |
| l = 0 (s-orbital) | m = 0 | 2 |
| l = 1 (p-orbital) | m = −1 | 6 |
| m = 0 | ||
| m = +1 | ||
| l = 1 (d-orbital) | m = −2 | 10 |
| m = −1 | ||
| m = 0 | ||
| m = +2 | ||
| m = +1 |
Magnetic quantum number of the potassium atom
We can also easily find out the magnetic quantum numbers from the electronic configuration data of an atom. For example, the electronic configuration of potassium (atomic number = 19) = 1s2 2s2 2p6 3s2 3p6 4s1.
Therefore, the last electron in the potassium atom resides in the 4s orbital. For s orbital, magnetic quantum number (m) = 0.
What are the 3 quantum numbers for the last electron in chlorine?
The electronic configuration of chlorine (atomic number 17) = [Ne] 3s2 3p5. Therefore, the last electron in chlorine resides in the 3p orbital, and for the 3p orbital, n = 3, l = 1. When l = 1, the magnetic quantum numbers (m) = −1, 0, or +1.
All the sub-orbital (px, py, pz) are degenerate, and they have the same energy. Therefore, the last electron of chlorine may reside in any of these degenerate sub-orbitals.
Spin Quantum Numbers
When the spectral lines of hydrogen, lithium, sodium, and potassium are observed by the spectrometer with high resolving power, each line of the spectral series consists of a pair of lines. These are known as doublets or double-line fine structures.
To account for these doublets, George Eugene Uhlenbeck and Samuel Abraham Goudsmit in 1925 suggested that when the electron moves around the nucleus in an orbit, it also rotates about its own axis either in a clockwise direction or in an anticlockwise direction.
Such spinning of the electron about its own axis adds to the angular momentum of an electron. It is called spin angular momentum, and it introduces a fourth or spin quantum number (s) of an electron in an atom.
How to find the spin quantum number?
Spin quantum number (s) can have two values, +½ and −½. The +½ value corresponds to the spinning of the electron in the clockwise direction, while the −½ value corresponds to the spinning in the anticlockwise direction. The clockwise and anticlockwise spinning are commonly represented by symbol ↑ and ↓ respectively.
From the above discussion, we can easily find out the spin quantum numbers of an electron in an atom, and it has values either +½ and −½.
Frequently Asked Questions
Which equation is used to derive quantum numbers?
Schrondinger equation is used to derive the first three quantum numbers (principal, azimuthal, and magnetic quantum numbers). However, the fourth or spin quantum number is an intrinsic property of an electron.
Why is the spin quantum number introduced?
When the spectral lines of various elements are observed by the spectrometer with high resolving power, each line of the spectral series consists of a pair of lines. To account for these doublets, George Eugene Uhlenbeck and Samuel Abraham Goudsmit in 1925 suggested that when the electron moves around the nucleus in an orbit, it also rotates about its own axis either in a clockwise direction or in an anticlockwise direction and introduced the spin quantum number.
How to find out the principal quantum number of the 11th electron of the sodium atom?
The atomic number of a sodium atom = 11. Thus, the 11 electrons in the sodium atom reside in the K shell (2 electrons), L shell (8 electrons), and the M shell (1 electron). Therefore, the principal quantum number of the last electron or 11th electron of the sodium atom = 3.
How to find out the azimuthal quantum number of the last electron of the vanadium atom?
The atomic number of vanadium = 23 and the electronic configuration = 1s2 2s2 2p6 3s2 3p6 4s2 3d3. Therefore, the last electron of the vanadium atom resides in the 3d orbital.
For 3d orbital, principal quantum shell (n) = 3 and azimuthal quantum number (l) = 2. Therefore, the azimuthal quantum number of the last electron of the vanadium atom = 2.
Why are quantum numbers important?
Quantum numbers or principal, azimuthal, magnetic, and spin quantum numbers in chemistry or physics is importent because they describe the size, shape, and orientation of an electron in an atom. However, the spin angular momentum introduces a fourth number or spin quantum number (s).